More than a decade ago, the scientists and engineers at PowerVision set out with simple yet lofty goals. They wanted to design an IOL that would provide true physiologic accommodation, allowing patients to see at both distance and near in a seamless fashion, and, as a requisite, yield 20/20 high-quality distance vision without induced aberration, glare, and dysphotopsia and at least 2.50 D of accommodation. The lens was to be based upon the natural physiologic mechanism of accommodation as we understand it, involving shape change, the ciliary muscle, and forward movement. It was determined that the lens would be designed for intracapsular placement both for physiologic reasons—so that it is sequestered and protected in the bag—and to allow conventional implantation techniques. These concepts have been realized in the development of a fluid-based accommodating IOL, the FluidVision AIOL.
LENS DESIGN AND IMPLANTATION
The FluidVision technology consists of a proprietary fluid-driven system housed within the lens. It is made of a hydrophobic acrylic material and has hollow haptics and a hollow central optic that contains an index refractive-matched silicone oil of ophthalmic grade. Small amounts of fluid—on the level of microliters—are transferred back and forth from the optic to the haptics to change the shape of the lens, thereby accommodating the way the crystalline lens accommodates.

The FruidVision lens.
The overall diameter of the FluidVision lens is 10 mm. The optic is 6 mm in diameter. The haptics are about 3-mL tall, and they are suspended on little struts that allow communication with the fluid in and out.
Initially, the FluidVision lens was implanted through a large incision. Over time, however, its implantation has been refined, and current work is being directed toward developing a better injector delivery device. Presently, the device is slightly under 4 mm but the next version will be 3.5 mm or less. Placing this fluid-filled lens into a small incision is no easy task. It involves, inside the body of the injector, placement of an OVD syringe, and it is visco-hydraulically compressed and injected into the capsular bag.
Unlike standard IOLs, the FluidVision lens is not a two-dimensional entity; there is girth and depth to the lens. The key is to get the initial haptic into the bag, and then the optic easily blossoms into the bag. The lens essentially auto-centers, and it really fills the bag. There is very little capsular change, which was an early concern.
STUDY RESULTS
The initial work on the lens was done, of course, on the bench. Blind eye studies began in 2009, followed by a pilot study of sighted eyes conducted by two surgeons in South Africa.
Follow-up data are now available up to 18 months for 20 pilot eyes implanted with the FluidVision lens. In these eyes, the mean level of distance acuity is actually better than 20/20. More importantly, near performance seems to be meeting our criteria. On the defocus data, the mean starts off slightly better than 3.00 D and then continues on. By way of pushdown accommodative measurements, we are seeing a mean of just better than 2.50 D of accommodative function. By way of pharmacologic testing using pilocarpine, these same patients are showing 2.50 D to 4.00 D of accommodative amplitude.
Eight of the 20 pilot patients had their second eye implanted with the FluidVision lens. Distance, intermediate, and near vision all increased. Most importantly, near vision improved from 20/32 and is now pushing 20/25.
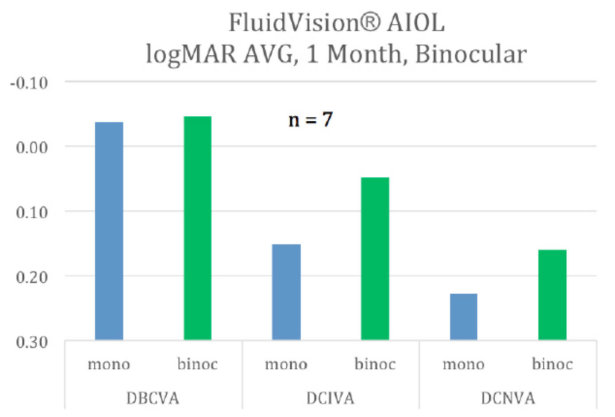
Distance, intermediate, and near vision increased in binocularly implanted eyes.
SUMMARY
Thus far, the FluidVision lens has been shown to provide very good acuity at distance and intermediate and, in terms of accommodative amplitude, to provide in the neighborhood of that minimum of 2.50 D. Binocularly implanted patients are showing even better function. The goal is to get additional patients implanted through multicenter studies, and then hopefully FDA trials will begin in the near future.


