Patients with keratoconus who present with decreased vision from cataracts present challenges in refractive outcomes. Factors such as ocular surface, irregular astigmatism, changes in keratometry, and contact lens history can all play a role in lens calculations leading to unanticipated refractive surprises. With careful consideration of the parameters for IOL calculations, as well as proper counseling, these patients can have excellent outcomes without the need for further surgery.
Screening Topography
Screening topography is important in catching keratoconus patients. The topography device does not need to measure more than the anterior cornea to screen for keratoconus; many image devices like the Atlas (ZEISS) can capture the anterior image and provide enough data for the surgeon to plan around the irregular astigmatism. However, if one does not do topography prior to surgery, one will not catch irregular astigmatism. Fortunately, we see surgeons using screening topography more often in cataract surgery evaluations.
Cornea: The Forefront of IOL Calculations
A pristine cornea is imperative for accurate lens calculations (Figure 1). Several studies have shown the more advanced the keratoconus, the less repeatable the keratometry values.1 Patients with keratoconus often have inconsistent keratometry values for several reasons. The timing of IOL calculations in patients with evident contact lens warpage varies based on the type of lens. In my practice, soft contact lenses need to be out for a full week prior to any measurements. A scleral lens on the other hand, which does not touch the cornea, would need to be out for 2 to 3 days prior to evaluation. This is very different than contact lenses, which cause more severe warpage, such as rigid gas permeable lenses, hybrid lenses, or even soft toric lenses. Although it is preferable for the patient to not wear these lenses for 2 weeks prior to evaluation, the evidence of warpage can continue for months, especially in those who have worn these lenses for years.
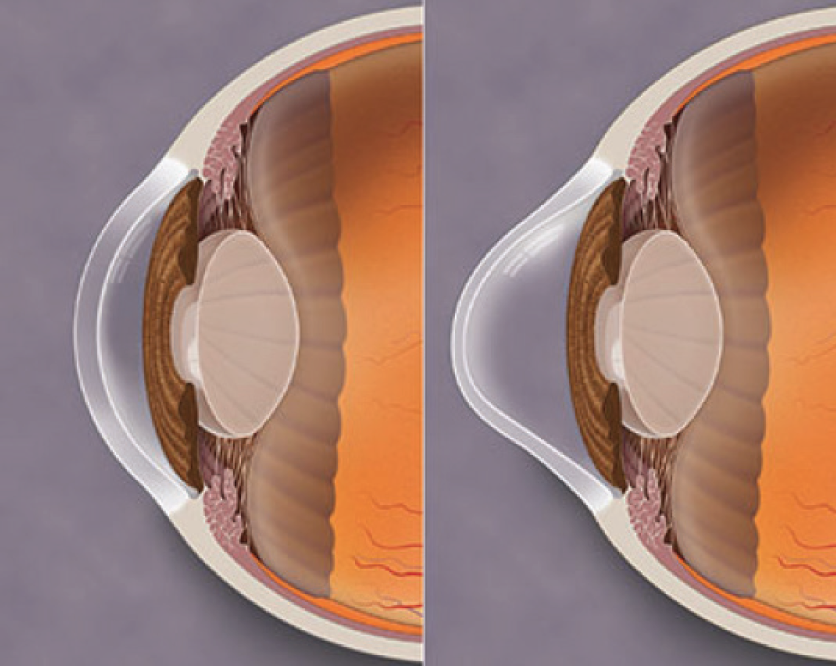
Figure 1. Normal cornea versus keratoconus.
Ocular Surface
Historically, keratoconus has been defined as a noninflammatory thinning of the cornea to set it apart from other diseases. However, we now know keratoconic eyes have increased inflammatory markers compared to normal eyes, as well as decreased sensitivity and tear breakup time. The more advanced the keratoconus, the more irregular the surface. Treatment of the condition is important prior to surgery. Older biometers are often unable to capture the keratometry in these eyes due to changing tear film. However, newer biometers like the IOLMaster 700 (ZEISS) can quickly capture images even in difficult patients. One study showed the average time to complete biometry measurements in both eyes was significantly shorter for the IOLMaster 700 compared with the Lenstar LS900 (Haag-Streit) (44.5 ±12.4 vs 168.8 ±67.2 seconds, P<0.001).2
Anterior vs. Posterior Cornea
Compared to previous methods of determining lens accuracy, new factors have emerged to determine more accurate IOL calculations with the anterior and posterior cornea gaining significant importance. Keratoconus is even more challenging in its measurements. The anterior chamber in eyes with moderate to advanced keratoconus is often irregular and deeper than in normal eyes. First-generation formulas do not include anterior chamber measurements at all, while newer-generation formulas may be relying on false information. Until recently, the SRK II formula may have been the most accurate in calculating IOL power for patients with mild keratoconus.3 It is likely that the keratometry values play a stronger role than the actual formula used, but new-generation formulas can help with these eyes with unusual anterior chamber depth.
In my practice, the first step is assessing if the patient presents with blurry vision from the keratoconus or from the cataract (or a third diagnosis). At times, this requires a contact lens over-refraction. The second step is to determine if the patient’s keratoconus is stable or progressive, which can be difficult in the setting of a worsening cataract. The third is to discuss the unique nature of this patient’s eyes. Patients with mild or moderate keratoconus can sometimes have unrealistic expectations that their surgery will be just like their family member’s or neighbor’s, with a 20/20 outcome.
Next is the tough part: picking the lens power. Here is where we see a clear pattern that the steeper the cornea values, the more hyperopic the outcome. This trend is repeated when utilizing keratometry values from devices such as Scheimpflug topography. Utilizing this trend, I pick the more myopic outcome based on the steep keratometry value. It is important to track outcomes and counsel the patients prior to surgery about this thought process.
Patient Case Study
To demonstrate, I saw a patient who had a known history of keratoconus. She had been tolerant of hard contact lenses in the past and still wore soft contact lenses for distance. She had fairly normal K readings (48.00 D) when we looked at those values alone, which is a little steep for astigmatism. One of the first things to do for a patient like this is to understand what is truly limiting vision. Is this a corneal issue? A cataract issue? A combination of the two? Once this is determined, then we can form a strategy for how to move forward. In our practice, we have the advantage of having a scleral trial set. Even though this patient had not worn her contact lenses in a while, we put the scleral trial frame on and over-refracted her. We saw limitations in her vision despite the contact lens and moved to better understand her vision goals. This patient loved to see things up close, and she loved to do needlework, so we determined her target was more of a true near. With our goal established, and knowing she was a keratoconus patient, the question then became, how much of a hyperopic outcome are we going to have? This is where the correct calculations become extremely important. The cornea was too irregular for corneal refractive surgery, so any negative outcome would require an IOL exchange if the calculations were off target.
Our target goal for these types of cases is ±0.50 D. This patient had a fairly normal to slightly steeper keratometry reading on biometry, and we anticipated a tendency for a hyperopic outcome, so we still aimed for some myopia but were confident that there weren’t many outliers in that group. We know that the steeper the cornea the more outliers, making repeatability of the keratometry extremely important. Before technology like the IOLMaster 700, having good repeatability in topographies or K readings was difficult. There were multiple variabilities not being taken into account, including ocular surface disease, instability of the tear film, or an irregular cornea. For this patient, we aimed for a myopic outcome and set our goal for -3.00 D, aiming at -3.50 D. We ended up with -2.75 D, which was a little on the hyperopic side but a fairly predictable (based on measurement) response for her cornea given the keratoconus (Table).
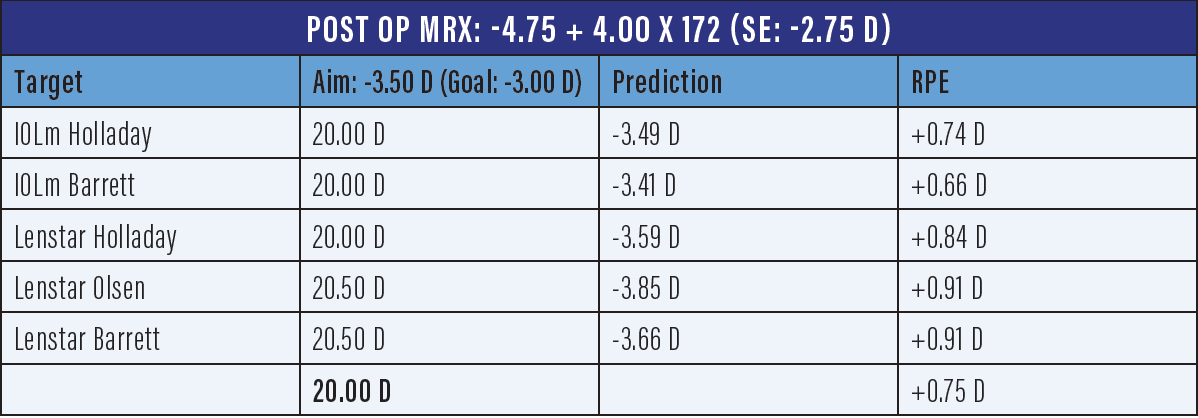
Table. For this patient, we aimed for a myopic outcome and set a goal of -3.00 D, aiming at -3.50 D. We ended up with -2.75 D.
Conclusion
In keratoconic eyes, optimizing IOL power calculations is certainly of vital importance toward the improvement of surgical outcomes and, ultimately, patient satisfaction. Keratoconus can cause multiple sources of keratometry and biometric errors. Addressing the cornea, utilizing the best technology, and considering taking multiple measurements can help optimize outcomes in these patients.
1. Hashemi H, Yekta A, Khabazkhoob M. Effect of keratoconus grades on repeatability of keratometry readings: comparison of 5 devices. J Cataract Refract Surg. 2015;41(5):1065-1072.
2. Passi SF, Thompson AC, Gupta PK. Comparison of agreement and efficiency of a swept source-optical coherence tomography device and an optical low-coherence reflectometry device for biometry measurements during cataract evaluation. Clin Ophthalmol. 2018;12:2245-2251.
3. Ghiasian L, Abolfathzadeh N, Manafi N, et al. Intraocular lens power calculation in keratoconus; a review of literature. J Curr Ophthalmol. 2019;31(2):127-134.




