
Keratoconus presents as a spectrum, from forme fruste keratoconus, with very little visual impact, to advanced disease. In severe cases, patients may experience enough loss of visual acuity to be considered disabled. Treatments like scleral contact lenses and keratoplasty can improve vision, while corneal cross-linking can prevent progression to severe disease. Keratoconus patients will all develop a cataract in their lifetime as well. For these patients, cataract surgery may be a viable surgical option with strong outcomes, but it presents a myriad of challenges in IOL calculation. Careful considerations must be made to the parameters of IOL calculations, including keratometry, effective lens position (ELP), axial length, ocular surface, irregular astigmatism, proper patient counseling, and more; these parameters are required to provide the best possible outcomes. A solid approach is understanding that the more severe the keratoconus, the more unanticipated refractive surprises you may experience, which leads to more challenging IOL calculations (Figure 1).
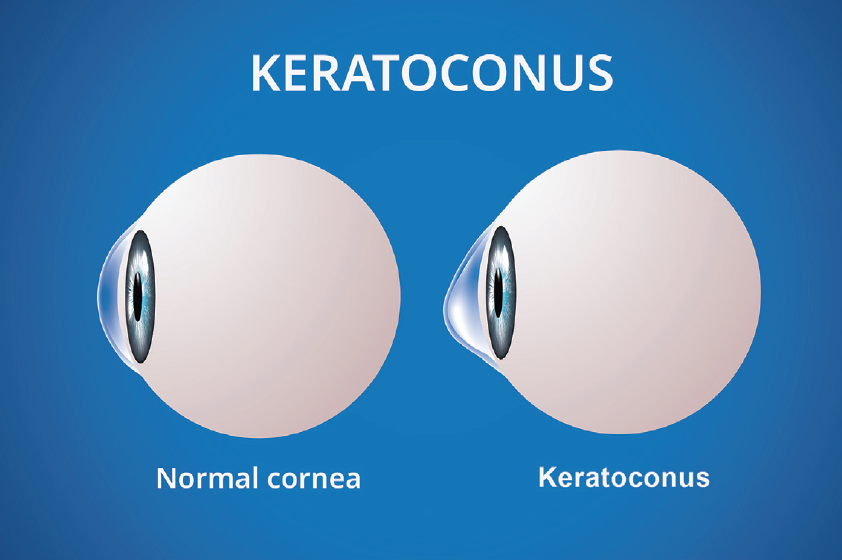
Figure 1. Normal eye versus an eye with keratoconus.
Inconsistent Keratometry
When planning cataract surgery for a patient with severe keratoconus, you should expect that the keratometry value (K) will be difficult to obtain. Several studies have shown the more advanced the keratoconus, the less repeatable the keratometry values.1 Today, many severe keratoconus patients wear contact lenses. This adds a level of compliance, requiring patients to “take a holiday” from wearing contact lenses for a set period of time. This process allows a reduction of warpage, in hopes of yielding repeatable K value readings. In my practice, and under normal conditions, soft contact lenses need to be out for a full week prior to taking any measurements. A scleral lens on the other hand, which does not touch the cornea, would need to be out for 2 to 3 days prior to evaluation. This is very different than contact lenses, such as hard lenses, rigid gas permeable lenses, and hybrid lenses, which cause more severe warpage. In my practice, even soft toric lenses require a contact lens holiday of at least 2 weeks. Patients with more advanced keratoconus (50+ K) who have been wearing contact lenses for years may have to remove those lenses for months, which is a difficult task for patients to undertake.
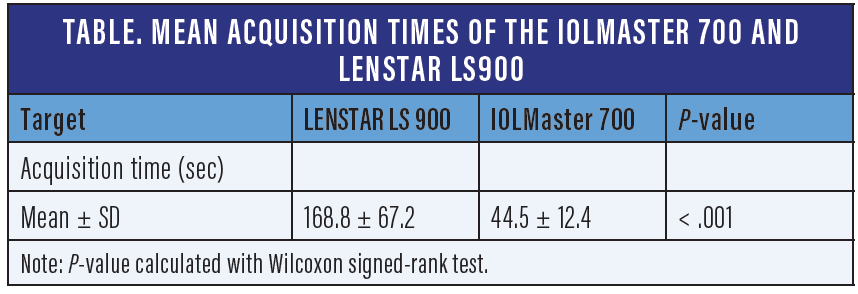
Often times, there are variations with sets of calculations taken within hours of each other due to an unstable tear film over the cone. This is when it’s helpful to have a topographer available, as well as access to biometry that takes measurements quickly, like the IOLMaster 700 (ZEISS). Older biometers are often unable to capture the keratometry in these eyes due to changing tear film. However, newer biometers, like the IOLMaster 700, can quickly capture images even in difficult patients. In fact, one study showed the average time to complete biometry measurements in both eyes was significantly shorter for the IOLMaster 700 compared with the Lenstar LS900 (Haag-Streit) (44.5 ± 12.4 vs 168.8 ± 67.2 sec, P < .001) (Table).2
Aiming for Myopia
In all cases of keratoconus, patient education and counseling are extremely important. In my practice, I approach these patients by explaining the importance of a contact lens holiday, and I try to gauge their comfort level with obtaining the most accurate IOL calculations. If the patient plans on continuing to wear contact lenses after cataract surgery, I aim to get 2 to 3 measurements 3 to 4 weeks apart during the patient’s contact lens holiday, or longer if the patient can tolerate it. If the patient cannot tolerate much time without wearing contact lenses, we can break up the holiday time periods or use the data we’ve collected and aim for some myopia. In these cases, it is easier for the contact lens specialist to fit a patient with a myopic, astigmatic correction rather than a mixed or hyperopic astigmatism. If the patient is amenable and prefers to move forward, rather than continuing the contact lens holiday, I aim for a more myopic lens and schedule a postoperative month 1 visit to see a contact lens specialist.
Anterior Chamber, ELP, and Axial Length
Severe keratoconus patients have unusual anterior chambers due to the extreme steepness and irregularity of the cornea. This makes it extremely difficult to measure. First-generation formulas do not include anterior chamber measurements at all, while newer-generation formulas may be relying on false information. It is important to use multiple IOL calculation methods for these eyes. In my practice, we try to use at least three different formulas using calculations from the Barrett Suite or Holladay Consultant, which are both available on the IOLMaster 700. Until recently, the SRK II formula may have been the most accurate in calculating IOL power for patients with mild keratoconus.3 It is likely that the keratometry values play a stronger role than the actual formula used, but new-generation formulas can help eyes with unusual anterior chamber depth. ELP is very difficult to understand in eyes with severe keratoconus. Often times the measurements of these severe cones make it difficult to estimate where the lens is going to sit. Severe keratoconic eyes also have longer axial lengths, so you have to keep that in mind when choosing formulas and nomograms.
Even when the correct IOL power is determined, the surgeon should be aware of the positive or negative sphericity of the IOL chosen. A keratoconic cornea is unique in its sphericity. Most corneas have increased positive spherical aberration over time, but the cone in the keratoconic cornea creates a negative spherical aberration, much like a cornea after hyperopic LASIK. Because most IOLs have a slight negative spherical aberration, one should consider choosing a true aspheric or positive spherical lens.
Patient Case Study
I had a 71-year-old patient who had very advanced keratoconus from wearing scleral lenses throughout the day. During a cataract evaluation, his keratoconus was very steep and diffusedly centered, with high myopia and a K value of 56.91 D (Figure 2). This patient never had a true manifest refraction that gave him accuracy of vision. It had always been scleral contact lenses and over-refractions for them. I went very myopic on this patient, aiming for a +2.00 D IOL power, which the IOLMaster 700 predicted as being almost -6.00 D. In cases like this, the contact lens specialist would much rather fit over a myopic, astigmatic prescription than a hyperopic astigmatism. It provides a lot more flexibility as far as how they vault and what diameter lens they choose. This patient came to me so thrilled, with 20/30 VA without correction and no longer needing to wear scleral lenses.
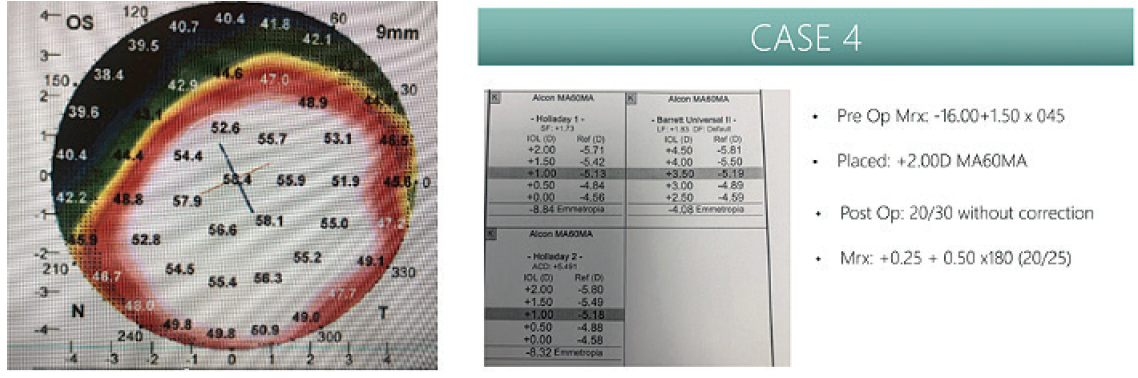
Figure 2. Patient case study. Ks: 55.94/57.90 (56.91 D) (left) and preoperative versus postoperative MRx (right).
Conclusion
In keratoconic eyes, optimizing IOL power calculations is certainly of vital importance to the improvement of surgical outcomes and, ultimately, patient satisfaction. Keratoconus can cause multiple sources of keratometry and biometric errors, especially in severe cases. To obtain the best outcomes, surgeons must take the extra time to address the complexities of the cornea, utilize the best technologies available, make careful preoperative measurements, adjust the refractive target based on calculations made with multiple formulas, and have a plan in place to address refractive surprises.
1. Hashemi H, Yekta A, Khabazkhoob M. Effect of keratoconus grades on repeatability of keratometry readings: comparison of 5 devices. J Cataract Refract Surg. 2015;41(5):1065-1072.
2. Passi SF, Thompson AC, Gupta PK. Comparison of agreement and efficiency of a swept source-optical coherence tomography device and an optical low-coherence reflectometry device for biometry measurements during cataract evaluation. Clin Ophthalmol. 2018;12:2245-2251.
3. Ghiasian L, Abolfathzadeh N, Manafi N, et al. Intraocular lens power calculation in keratoconus; a review of literature. J Curr Ophthalmol. 2019;31(2):127-134.



