An estimated 30% of patients undergoing cataract surgery have more than 1.00 D of corneal astigmatism,1 with 10% having more than 2.00 D.2 A recent survey completed by the American Society of Cataract and Refractive Surgery (ASCRS) found that toric IOLs were placed in only 9% of all cataract procedures performed.3 Patient expectations for spectacle independence are on the rise. Ensuring accurate toric IOL alignment is critical to this process but adds significant complexity to both the preoperative preparation and intraoperative steps, making refractive cataract surgery increasingly challenging.
PREOPERATIVE PLANNING
Preoperative planning for toric IOL placement requires accurate measurement of the magnitude, axis, and regularity of corneal astigmatism. Currently available methods of astigmatism measurement include manual, automated, and simulated keratometry, with automated keratometry having the greatest repeatability.4 We recommend comparing data from more than one modality, looking for agreement to confirm axis and magnitude and thereby minimizing errors.5 This is also necessary to accurately determine the appropriate spherocylindrical lens power prior to selecting a toric IOL, which can be streamlined by using the online calculators provided by the individual IOL manufacturers or third-party calculators. The Barrett calculator has the distinct advantage of accounting for the posterior corneal surface and, in a study by Abulafia et al,6 was shown to be the best predictor of residual corneal cylinder.
The most common cause of postoperative refractive errors after toric IOL placement is inaccurate preoperative calculations, highlighting the importance of accurate preoperative planning and measurements. One of the most common errors in the measurement of astigmatism is irregularity of the corneal surface from ocular surface disease.7 It is our customary practice to treat ocular surface disease aggressively prior to determining which lens type is best suited for cataract patients. Tomography methods such as the Pentacam (Oculus Optikgeräte) can measure the posterior cornea and thereby calculate total corneal cylinder power, which has also been shown to improve postoperative outcomes.8
TORIC ALIGNMENT
There are three major steps to aligning the toric IOL to the appropriate axis:
(1) determination of corneal astigmatism (magnitude and axis), (2) marking of the reference angle, and (3) proper alignment of the lens. Correct surgical alignment of toric IOLs requires accurate preoperative and/or intraoperative determination of the steep axis. Failure to accurately align the orientation marks of the toric IOL (which is 90° opposite the steep power axis) with the steep axis of the cornea will lead to not only a decreased amount of cylindrical correction in the desired axis but also the possibility of induction of new astigmatism based on vector analysis. A sinusoidal relationship exists between misalignment and the induced cylinder. Small misalignments cause a 3% loss of intended astigmatic correction per degree of misalignment, while a 30° misalignment causes effectively complete nullification of the intended astigmatic correction.9
MANUAL ALIGNMENT
Traditional preoperative marking of the horizontal meridian is generally performed using one of a number of manual devices, including bubble markers, pendular markers, tonometer-based markers, and manual slit lamp marking. About 84% of surgeons utilize manual marking for toric alignment.3 Additionally, most patients have 2° or more of cyclotorsion when changing from seated to supine position. To avoid torsion-induced errors, most surgeons perform preoperative marking of the horizontal meridian while the patient is seated and fixating at a distance target.9
The next step is the intraoperative marking of the steep axis. This is traditionally performed using a manual graded measuring instrument such as a Mendez marker or LRI marker to measure and mark the location of the steep axis relative to the preoperatively marked horizontal meridian. Once the steep axis is marked, the toric IOL can be aligned accordingly. Advances in femtosecond laser cataract surgery technology have made it possible to automate the intraoperative marking of the steep axis using a small intrastromal laser mark on the peripheral cornea.10
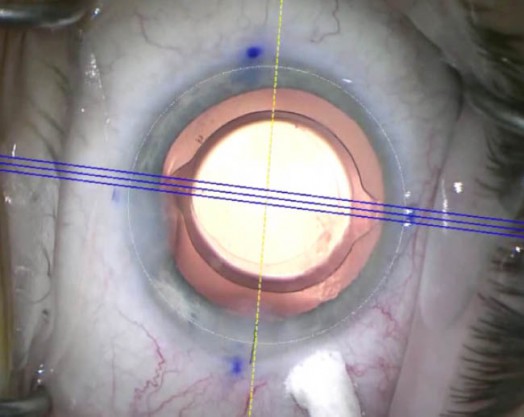
Figure 1.Intraoperative toric IOL alignment using the Callisto. The reference alignment guide is shown in yellow, and the toric alignment guide is shown as a triple blue line. Note the difference in reference between manual marking (limbal ink marks) and automated reference as captured by the Callisto.
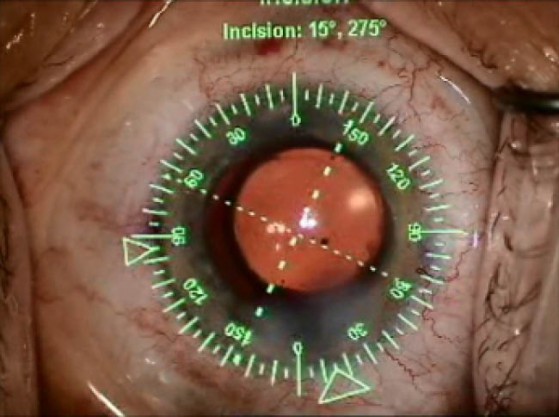
Figure 2.Intraoperative image from the Verion system, with identification of the steep axis for toric IOL alignment.
IMAGE-GUIDED ALIGNMENT
Image-guided alignment systems include the Callisto (Carl Zeiss Meditec; Figure 1), Verion (Alcon; Figure 2), and TrueVision (TrueVision Systems). These systems use preoperative anterior segment photographs to map the desired corneal steep axis relative to reference limbal vessels.11 Intraoperatively, the preoperative reference image is aligned to similar intraoperative images, allowing the system to project the steep axis through the surgical microscope to guide IOL alignment.
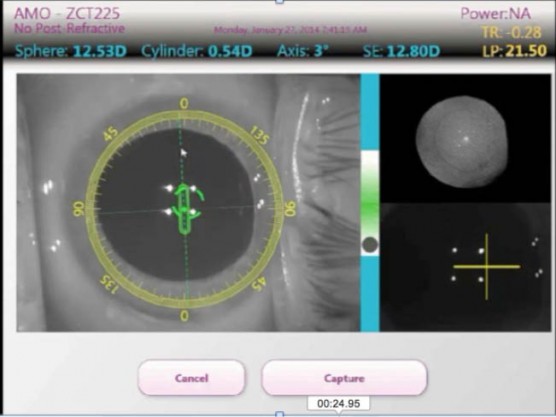
Figure 3. ORA intraoperative aberrometery showing real-time astigmatic measurement (magnitude and axis).
ABERROMETRY-GUIDED ALIGNMENT
Aberrometry-guided alignment systems can aid in reconfirming preoperative measurements, offering another data point with the goal of obtaining a higher degree of precision and minimizing alignment error. Aberrometry-guided alignment has the added benefit of confirming spherical power. Additionally, aberrometry-guided alignment methods take into account the posterior corneal astigmatism. There are two systems commercially available on the US market: the ORA (Alcon; Figure 3) and Holos (Clarity Medical Systems,). The ORA system uses Talbot Moiré interferometry to develop a fringe pattern from which measured aberrations are converted to refractive values. The ORA features a large dynamic range of -5.00 D to +20.00 D and enables real-time surgical course correction. The Holos, on the other hand, uses a wavefront aberrometer to provide continuous refractive error readout throughout the procedure.12
Intraoperative aberrometry is most reproducible when measurements are conducted under aphakic conditions in the presence of viscoelastic.13 After placement of an IOL, immediate pseudophakic refraction can be used to align toric IOLs, determine residual refractive error, and even guide placement of limbal relaxing incisions for any residual cylinder. A recent study found that intraoperative aberrometry led the surgeon to change the preoperatively selected sphere and cylinder powers in 25% and 24% of cases, respectively.14 Further, patients in whom intraoperative aberrometry was used were 2.4 times more likely to have less than 0.50 D of residual postoperative astigmatism compared with manual techniques.14 Aberrometry can both identify the desired final axis and guide the intraoperative rotation in real time, although preoperative planning is still recommended. The Berdahl/Hardten calculator is an excellent resource to determine the rotation necessary to realign a misaligned toric IOL.15,16
Factors that may compromise intraoperative aberrometry results include corneal opacities, dry ocular surfaces, insufficient intraocular pressures (ideal >25 mm Hg), or errors induced by the eyelid speculum.17 Aberrometry also requires a compliant patient with proper fixation. Intraoperative aberrometry can also be inaccurate if preoperative biometry is greatly erred. Even with these potential problems, intraoperative aberrometry still has many advantages over manual toric alignment.
ADDITIONAL OPERATIVE CONSIDERATIONS
Regardless of which alignment method is used, post-alignment operative techniques can greatly affect the final rotational position of toric IOLs. The most common operative cause for toric IOL misalignment is incomplete removal of viscoelastic, especially if left behind the IOL.15 Additionally, when removing viscoelastic, the toric IOL may have a tendency to rotate clockwise. This potential error can be avoided by using a second instrument to fix the toric IOL to the posterior capsule while removing the viscoelastic, thus preventing rotation.18,19
CONCLUSION
Toric IOL alignment techniques are an increasingly essential skill for refractive cataract surgeons wishing to better address this untapped market. Manual, image-guided, and aberrometry-guided alignment techniques have their benefits and limitations, but all yield excellent postoperative results, especially when compared with non-toric IOL placement in a patient with preexisting corneal astigmatism.
1. Hoffer KJ. Biometry of 7,500 cataractous eyes. Am J Ophthalmol. 1980;90:360-368.
2. Hoffmann PC, Hutz WW. Analysis of biometry and prevalence data for corneal astigmatism in 23,239 eyes. J Cataract Refract Surg. 2010;36:1479-1485.
3. American Society of Cataract and Refractive Surgery. Clinical Survey. November 2015. https://www.eyeworld.org/supplements/ASCRS-Clinical-Survey-2015.pdf. Accessed February 12, 2016.
4. Mehravaran S, Asgari S, Bigdeli S, et al. Keratometry with five different techniques: a study of device repeatability and inter-device agreement. Int Ophthalmol. 2014;34:869-875.
5. Browne AW, Osher RH. Optimizing precision in toric lens selection by combining keratometry techniques. J Refract Surg. 2014;30:67-72.
6. Abulafia A, Barrett GD, Kleinmann G, et al. Prediction of refractive outcomes with toric intraocular lens implantation. J Cataract Refract Surg. 2015;41:936-944.
7. Montés-Micó R. Role of the tear film in the optical quality of the human eye. J Cataract Refract Surg. 2007;33(9):1631-1635.
8. Abulafia A, Hill WE, Franchina M, Barrett GD. Comparison of methods to predict residual astigmatism after intraocular lens implantation. J Refractive Surg. 2015;31(10):699-707.
9. Ma JJ, Tseng SS. Simple method for accurate alignment in toric phakic and aphakic intraocular lens implantation. J Cataract Refract Surg. 2008;34:1631-1636.
10. Montes de Oca I, Kim EJ, Wang L, et al. Accuracy of toric intraocular lens alignment and predicted residual astigmatism using a 3D computer-guided visualization system in femtosecond laser-assisted cataract surgery. Invest Ophthalmol Visual Sci. 2015;56:1913.
11. Cha D, Kang SY, Kim SH, et al. New axis-marking method for a toric intraocular lens: mapping method. J Refract Surg. 2011;27:375-379.
12. Krueger RR, Shea W, Zhou Y, et al. Intraoperative, real-time aberrometry during refractive cataract surgery with a sequentially shifting wavefront device. J Refract Surg. 2013;29:630-635.
13. Huelle JO, Katz T, Druchkiv V, et al. First clinical results on the feasibility, quality and reproducibility of aberrometry-based intraoperative refraction during cataract surgery. Br J Ophthalmol. 2014;98:1484-1491.
14. Hatch KM, Woodcock EC, Talamo JH. Intraocular lens power selection and positioning with and without intraoperative aberrometry. J Refract Surg. 2015;31:237-242.
15. Lockwood JC, Randleman JB. Toric intraocular lens rotation to optimize refractive outcome despite appropriate intraoperative positioning. J Cataract Refract Surg. 2015;41:878-883.
16. Berdahl JP, Hardten DR. Residual astigmatism after toric intraocular lens implantation. J Cataract Refract Surg. 2012;38:730-731.
17. Stringham J, Pettey J, Olson RJ. Evaluation of variables affecting intraoperative aberrometry. J Cataract Refract Surg. 2012;38:470-474.
18. Garzon N, Poyales F, de Zarate BO, et al. Evaluation of rotation and visual outcomes after implantation of monofocal and multifocal toric intraocular lenses. J Refract Surg. 2015;31:90-97.
19. Thulasi P, Khandelwal S, Randleman J. Intraocular lens alignment methods. Curr Opin Ophthalmol. 2015;26:1-11.


