Quality of view, safety, and efficiency are just a few of the reasons single-use optics are likely to become prevalent in future ophthalmic practice. The technique of gonioscopy, commonly used in both the operating room and clinic, requires a lens to overcome the total internal reflection of the cornea in order to image the anterior chamber and angle. Traditionally, gonioscopy lenses have been reusable, which means they must be cleaned and disinfected or sterilized after use, and repaired or replaced when damaged. Due to the limitations of reusable equipment, single-use devices have been gaining popularity in many medical fields, including ophthalmology. For example, cataract surgery may be trending toward single-use instruments, particularly outside of the United States.1
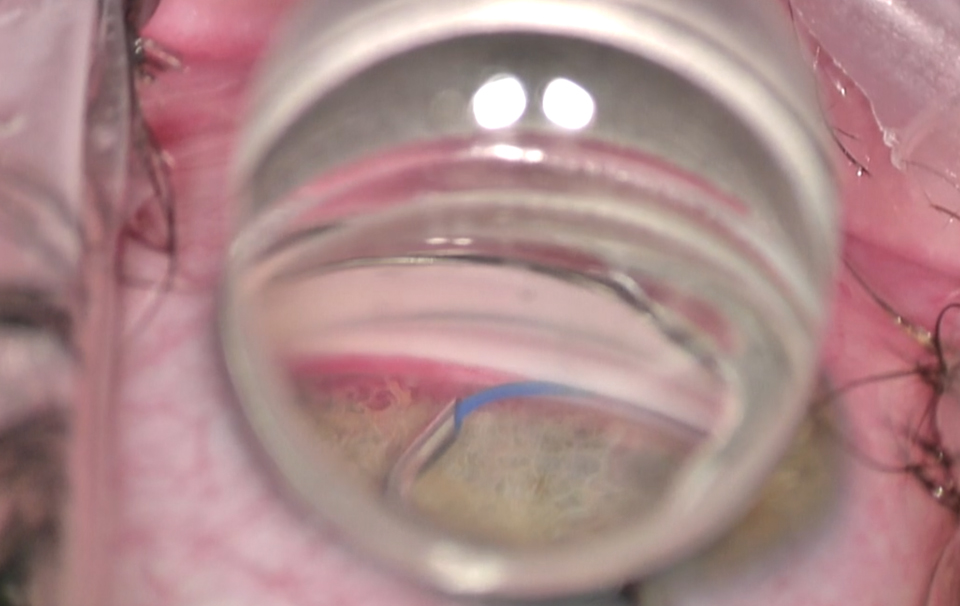
Figure 1 | Intraoperative view of an ab interno trabeculotomy with Katena’s single use gonioprism.
(Courtesy of Arsham Sheybani, MD)
Our experience with single-use gonioprisms (Katena Products) in the operating room has been excellent. These lenses bear significant similarities in design and use to the widely available reusable direct view surgical prisms, such as the Swan-Jacob lens (Ocular Instruments), except they have the advantage of a vault at the base of the optic, which allows easier instrument access. In my experience, single-use optics provide a view comparable to the standard nondisposable version. In fact, at times, imaging can be better because the one-time use ensures that the lenses do not have scratches, water marks, or cracks that cause unwanted glare and inhibit the view (Figure 1).

Figure 2 | Single-use lenses for gonioscopy: four-mirror (A), three-mirror (B), and single-mirror (C) lenses.
(Courtesy of Katena Products)
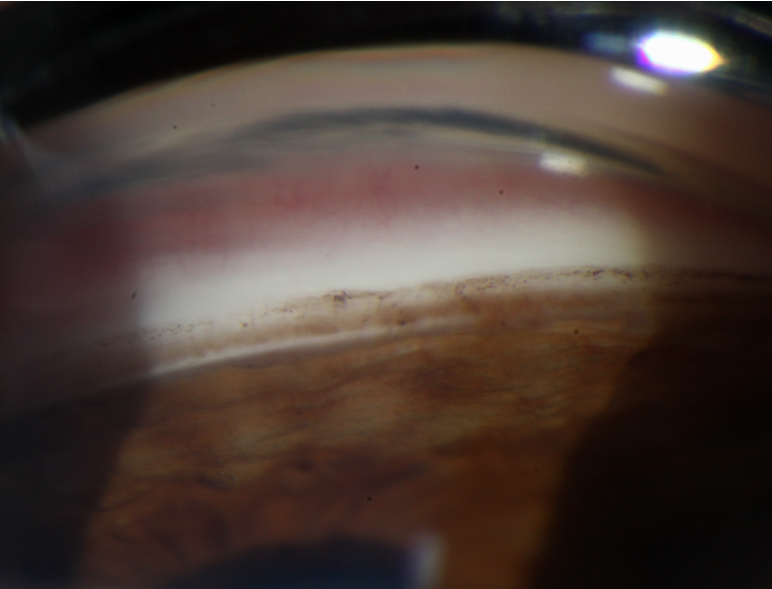
Figure 3 | Slit-lamp photo of the angle using Katena’s four-mirror lens.
(Courtesy of Arsham Sheybani, MD)
Disposable gonioscopy lenses have also recently become available for the clinic. Single-mirror, four-mirror (with and without a handle), and three-mirror lenses (Figure 2) are available and all provide surprisingly good optics. The single-mirror lens (Figure 3) has all of the necessary coatings for imaging as well as for selective laser trabeculoplasty or argon laser trabeculoplasty. The four-mirror lens is not suitable for lasering but has the advantage of not requiring contact fluid, which is convenient for both doctor and patient.
At approximately $25 per gonioprism, the cost relating to a surgical procedure is practical. The clinical lenses are priced at around $10 each, which, for diagnostic examination in a private practice setting, may be less attractive, as this represents about half of the CMS reimbursement. However, single-use lenses could be advantageous in the clinic in certain circumstances, particularly when Joint Commission standards must be met, for complicated cases where risk of disease transmission may be higher, or when the reusable lenses have been lost or damaged.
In terms of influencing how we practice ophthalmology, single-use lenses have many advantages, some obvious and others less so. In addition to no longer needing to clean, disinfect, or sterilize lenses, there is the potential for increased efficiency; staff are free for other duties, and the cost of disinfection/sterilization is removed, as is staff training for reprocessing procedures. Patient safety may also be affected, as there is no possibility of spreading infection between patients.
Another aspect of safety relates to the manner in which surgical instruments are reprocessed. In the developed world, some facilities’ sterilization processes are based on ethylene oxide (EtO) gas systems. EtO has been linked to chromosomal damage and is thought to be related to increased numbers of cancer deaths among workers exposed to the substance.2 Additionally, EtO is considered a fire and explosion hazard, and the product has been linked to destruction of the ozone.3 Given these deleterious effects, avoiding EtO would be beneficial to workers as well as the environment.
Our experiences to date suggest that disposable gonioscopy lenses are an excellent alternative to reusable lenses—and they are likely to become more widely adopted, especially with the projected increase in angle-based procedures.
1. Amzallag T. Single-use instruments becoming better options. Cataract & Refractive Surgery Today Europe. June 2010.
2. Landrigan PJ, Meinhardt TJ, Gordon J, et al. Ethylene oxide: an overview of toxicologic and epidemiologic research. Am J Ind Med. 1984;6(2):103-115.
3. Ethylene oxide sterilization: how hospitals can adapt to the changes. Health Devices. 1994;23(12):485-492.

