Independent series supported with
advertising by

Surgically induced corneal changes for the correction of astigmatism have been performed for more than 100 years. These treatment approaches were reevaluated in the early 90s and then recently further refined with the introduction of the femtosecond laser. Although arcuate incisions can be performed with a blade, I think many surgeons would agree that the laser is more precise for various reasons. Several factors directly affect the results; incision depth, width, architecture, radius from vertex (or conversely limbus), and axis of orientation are a few of the noteworthy variables.1 In contrast, corneal biomechanics have yet to be fully elucidated.2
KEY CONSIDERATIONS
When getting started with corneal astigmatic incisions, there are several important considerations to keep in mind. Presuming there is no need to further refine the spherical component, then it is safe to proceed. Secondly, a clear understanding of your surgically induced astigmatism (SIA) is imperative. This calculation is influenced by the impact your primary and secondary incisions have on the postoperative corneal shape. Understanding your SIA requires confidence in your reproducibility of the surgical technique and fidelity in postoperative data acquisition for comparison, usually with your preferred topographer or keratometer.
Total corneal power calculations are facilitated by devices such as the Galilei Dual Scheimpflug Analyzer (Ziemer Ophthalmic Systems), Cassini (i-Optics), and Pentacam (Oculus). My outcomes have improved with my understanding of the contribution from the posterior cornea.3 There are several well-established nomograms for both blades and laser from which to choose, and strict adherence during early adoption is recommended.4-6
REPRODUCIBILITY
At the risk of overstating the obvious, the advantage of the laser when compared with a bladed incision is the reproducibility. For example, appreciation of the incision depth at a micron level if the intention is to then deliver a 90% dissection is more likely to be regularly achieved with the femtosecond laser (Figure 1). Another distinct advantage provided by the laser is the opportunity to exploit the photodisruptive effect later. Whereas the blade will leave an incision that penetrates the epithelium, a titratable laser-assisted arcuate incision will often have an effect with the intrastromal dissection alone.
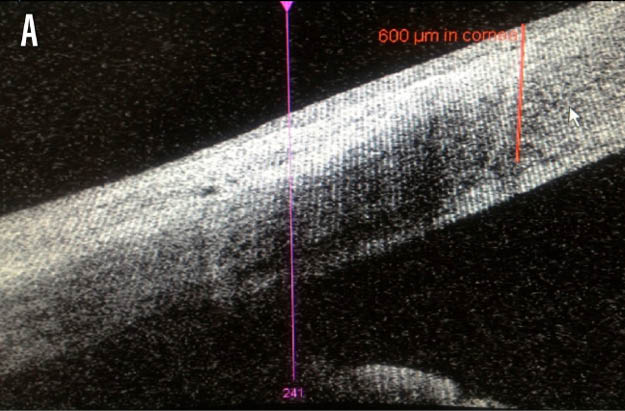
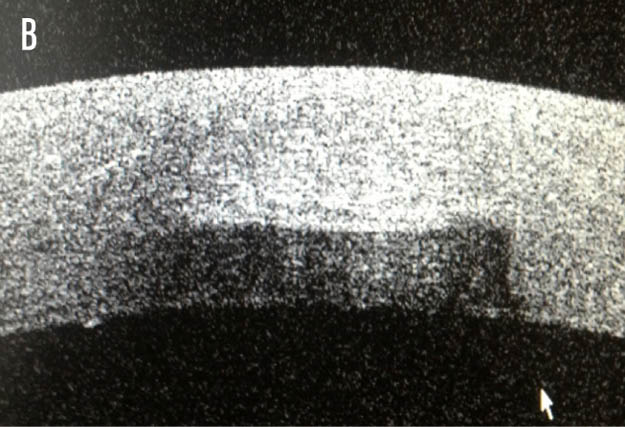
Figure 1. Anterior segment OCT cross-sectional images of the axial (A) and sagittal (B) planes, illustrating a depth of 600 μm.
REFINING OUTCOMES
Personally, I have made every effort to avoid deviating from this plan of tight adherence to a set nomogram. As a result, this has enabled me to become more assertive about addressing lower levels of astigmatism. I prefer to reserve corneal arcuate incisions for less than 1.50 D of with-the-rule stigmatism and less than 1.25 D of against-the-rule astigmatism. I will consider treating when there is the expected to be more than 0.50 D, based on total corneal calculations.
I often evaluate the impact of the arcuate incisions as part of my intraoperative management using aberrometry. However, I have found that, as my own nomogram has been refined, the need for dissection of the tissue bridges created with the femtosecond laser has decreased. I like to review the outcomes in a single-angle polar plot and consider the difference vector.
In other words, it is not good enough to know the absolute value of astigmatism following the surgical intervention; the angular change is equally as important. Should the need arise, typically because of mild undercorrection, it is relatively straightforward to titrate the correction by opening a laser arcuate incision at the slit lamp based on the refraction/topography. Keeping this in mind, standardizing treatment helps reduce variability, and success has followed.
CONCLUSION
Whether astigmatism is to be managed as part of a lens-based procedure or as a standalone intervention, I am confident that the millennial refractive specialist can achieve rapid adoption and early success with limbal relaxing incisions or arcuate incisions as a surgical technique.
1. Nevyas-Wallace A, Studer HP, Roberts CJ. Effect of novel architecture for femtosecond laser astigmatic keratotomy on induced aberration on patient-specific computation modeling. Invest Ophthal Vis Sci. 2014;55:1553.
2. Denoyer A, Ricaud X, Van Went C, Labbe A, Baudouin C. Influence of corneal biomechanical properties of surgically induced astigmatism in cataract surgery. J Cataract Refract Surg. 2013;39:1204-1210.
3. Koch DD, Ali SF, Weikert MP, Shirayama M, Jenkins R, Wang L. Contribution of posterior corneal astigmatism to total corneal astigmatism. J Cataract Refract Surg. 2012;38:2080-2087.
4. Abbey A, Ide T, Kymionis GD, Yoo SH. Femtosecond laser-assisted astigmatic keratotomy in naturally occurring high astigmatism. Br J Ophthalmol. 2009;93:1566-1569.
5. Day AC, Lau NM, Stevens JD. Nonpenetrating femtosecond laser intrastromal astigmatic keratotomy in eyes having cataract surgery. J Cataract Refract Surg. 2016;42:102-109.
6. Nichamin LD. Astigmatism management for modern phaco surgery. Int Ophthalmol Clinic. 2003;43:53-63.



