A 19-year-old woman presented with a 2-week history of a corneal opacity and photophobia in her right eye, which appeared 3 days after she underwent LASIK at another practice. The LASIK surgeon had treated the patient for presumed diffuse lamellar keratitis (DLK) that continued to worsen despite treatment.
Upon presentation, the patient was using difluprednate 0.05% and gatifloxacin 0.5% four times daily in the right eye. Her visual acuity was 20/70 OD and 20/20 OS. Slit-lamp examination showed stellate infiltration with slightly greater than 1 mm in the superior paracentral cornea within the LASIK interface and an overlying 1.5 mm epithelial defect. Anterior segment optical coherence tomography (OCT) showed arborizing linear elements present behind the cornea that extended into the anterior chamber. Confocal microscopy confirmed a filamentous fungal infiltrate (Figure 1).
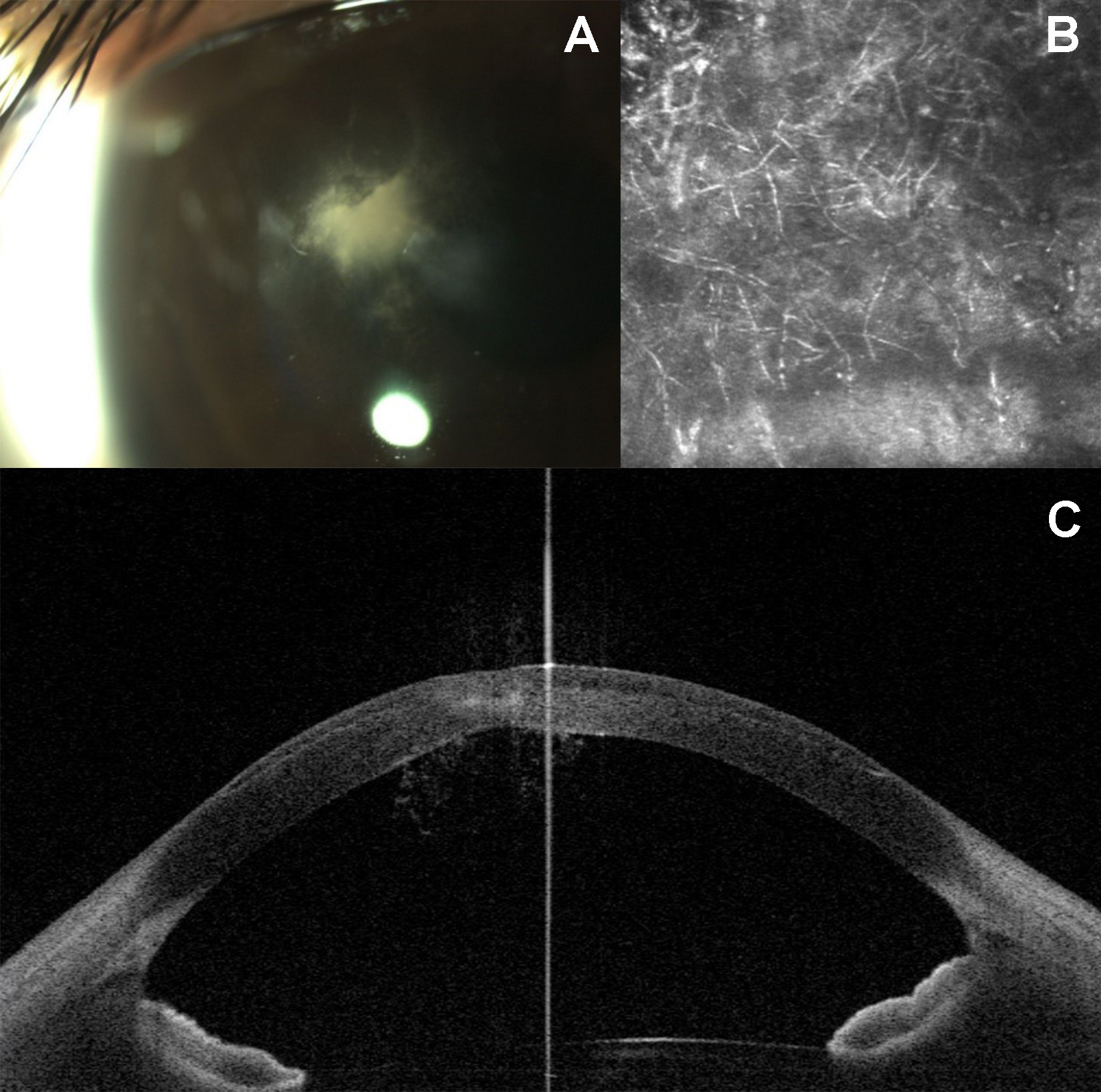
Figure 1 | Right corneal infiltrate in the LASIK interface upon initial presentation (A). Anterior segment OCT imaging demonstrating invasion of hyphal elements (B). Confocal imaging demonstrating diffuse hyphal elements in the corneal infiltrate (C).
On day 1 of presentation, natamycin 5% four times daily was started, and the difluprednate was discontinued. With the addition of natamycin, the patient subsequently developed diffuse DLK, and her vision decreased to hand motion at 3 feet on day 3. Due to extreme discomfort, prednisolone acetate 1% four times daily was added despite concern for fungal infection, in addition to oral fluconazole 200 mg and atropine 1% four times daily.
Despite clinical improvement of the DLK, the corneal infiltrate worsened, and the retro-corneal hyphal elements started to extend into the angles on days 4 to 8 (Figure 2). With the worsening state of the anterior chamber and concern for endophthalmitis, an anterior chamber paracentesis was performed on day 8 to obtain a culture sample, and 100 µg of voriconazole were injected into the anterior chamber.
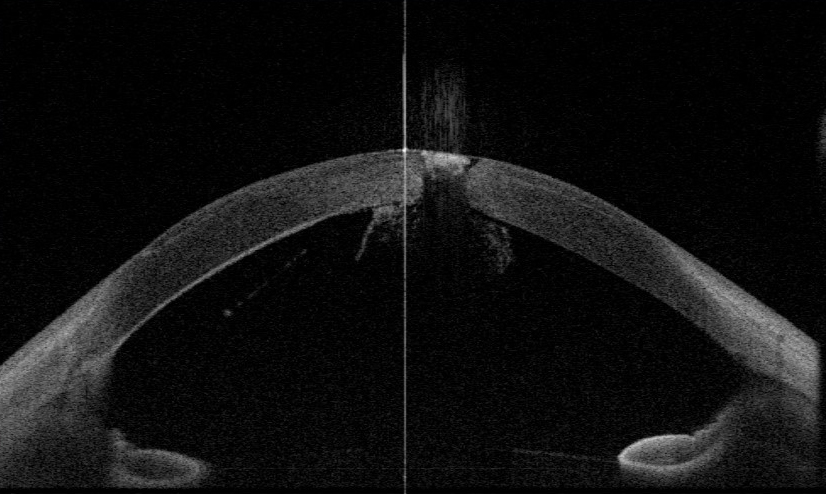
Figure 2 | Demonstration of the hyphae extending to the angle on anterior segment OCT.
Despite aggressive intervention, the patient worsened and presented with a corneal perforation on day 10. An urgent therapeutic 9.0-mm penetrating keratoplasty was performed. Upon removal of the cornea, multiple hyphal elements were attached to the cornea, angle, and iris; each adhesion was manually removed with forceps. The anterior chamber was irrigated with 20 mL of voriconazole 10 mg/mL, and 1 mL of fluconazole 2% intravenous solution was injected subconjunctivally 360 around the limbus.
After surgery, the patient was continued on atropine 1% and gatifloxacin 0.5%, with added cyclosporine 0.5% to prevent graft rejection. She was also continued on 200 mg twice daily of fluconazole and voriconazole 1% drops until 3 months postoperatively. The patient recovered without recurrence of the infection; at 3 years’ follow-up, her vision had improved to 20/20 with rigid gas permeable lens correction.
DISCUSSION
Post-LASIK infection is a rare complication of refractive surgery, occurring at a reported rate of 0.03%.1 TheIt was found that the risk of microbial keratitis is actually greater with more than 1 year of contact lens use than after LASIK surgery.2 As these infections are exceedingly rare, many times post-LASIK infections can be mismanaged, as the approach is different from a traditional corneal ulcer.
Upon initial suspicion of an infection after LASIK, it is recommended to elevate the flap and culture and scrape the underlying growth. It is not advisable to begin empiric therapy right away.3 If the infection is thought to have started within 2 weeks of the LASIK procedure, treatment is recommended with gatifloxacin 0.3% or moxifloxacin 0.5% every 5 minutes for three doses and then every 30 minutes, interchanging with cefazolin 50 mg/mL every 30 minutes.3 If onset is greater than 2 weeks after LASIK, amikacin 35 mg/mL every 30 minutes should replace cefazolin in the aforementioned regimen.3 In all cases, use of corticosteroids should be terminated. This patient’s initial prescription of difluprednate 0.05% for 2 weeks by an outside physician likely exacerbated the presenting fungal infection.
Fungal keratitis has the ability to penetrate an intact cornea and go through Descemet membrane, as was the case with our patient.4,5 If conservative measures are not effective, therapeutic penetrating keratoplasty must be performed to remove the source of infection. This case illustrates that, with aggressive intervention, positive outcomes can occur, even in the face of extensive fungal extension into the anterior chamber.
Supported in part by the Hermann Eye Fund.
1. Solomon R, Donnenfeld ED, Azar DT, et al. Infectious keratitis after laser in situ keratomileusis: results of an ASCRS survey. J Cataract Refract Surg. 2003;29:2001-2006.
2. Masters J, Kocak M, Waite A. Risk for microbial keratitis: Comparative metaanalysis of contact lens wearers and post-laser in situ keratomileusis patients. J Cataract Refract Surg. 2017;43:67-73.
3. Donnenfeld ED, Kim T, Holland EJ, et al; American Society of C, Refractive Surgery Cornea Clinical Committee. ASCRS White Paper: Management of infectious keratitis following laser in situ keratomileusis. J Cataract Refract Surg. 2005;31:2008-2011.
4. Naumann G, Green WR, Zimmerman LE. Mycotic keratitis. A histopathologic study of 73 cases. Am J Ophthalmol. 1967;64:668-682.
5. Taylan Sekeroglu H, Erdem E, Yar K, Yagmur M, Ersoz TR, Uguz A. A rare devastating complication of LASIK: bilateral fungal keratitis. J Ophthalmol. 2010;2010:450230.




