
A previously healthy 14-year-old was referred for bilateral eye redness and irritation. On exam, BCVA was 20/60 in the right eye and 20/30 in the left eye. Intraocular pressures were normal bilaterally. Slit-lamp examination revealed 4+ anterior chamber cells and posterior synechiae in the right eye and 3+ anterior chamber cells in the left eye. Dilated fundus exam was normal in both eyes.
After consultation with a pediatric rheumatologist and a negative targeted laboratory workup, a diagnosis of bilateral idiopathic chronic anterior uveitis was made. Initial management was with topical prednisolone acetate. After 1 month, weekly 25 mg subcutaneous methotrexate was added. Subsequently, 40 mg adalimumab (Humira, AbbVie) every 2 weeks was added and then increased to weekly dosing.
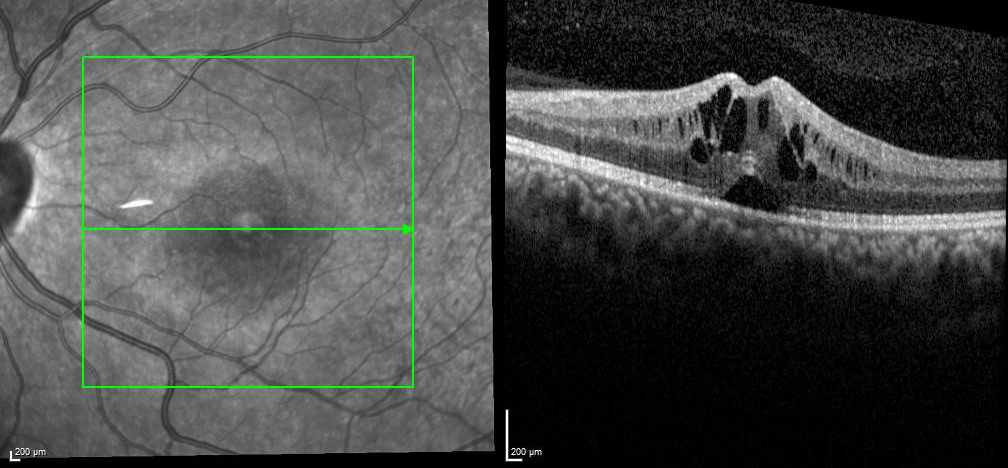
Figure 1 | Central foveal thickness before tocilizumab was 521 µm.
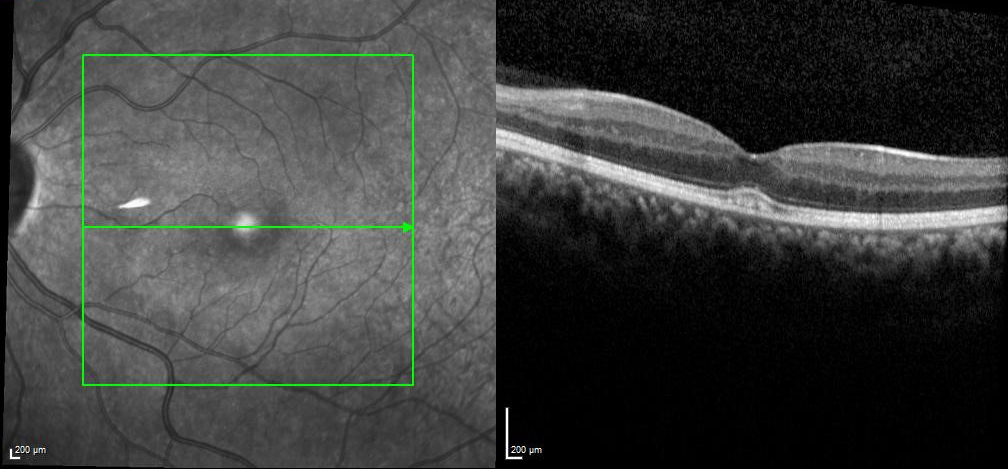
Figure 2 | After approximately two doses of tocilizumab, central foveal thickness was 283 µm.
Despite maximal dual therapy, adequate steroid-sparing control was not achieved, and the patient developed bilateral steroid-induced glaucoma and cataracts. She was switched from adalimumab to 5 mg/kg infliximab (Remicade, Janssen) every 4 weeks, which achieved rapid quiescence. The patient underwent glaucoma anterior tube shunt placement, followed by uncomplicated cataract surgery with IOL placement in both eyes.
A few months after cataract surgery, she developed bilateral cystoid macular edema (CME). Dilated examination revealed no intermediate uveitis, and a fluorescein angiogram revealed no vasculitis. Findings were initially attributed to pseudophakic CME and treated with a combination of topical NSAID and steroids, with initial resolution.
Attempts to taper topical steroids were unsuccessful, with recurrence of anterior uveitis and CME. Infliximab was titrated up from 5 mg/kg to 15 mg/kg at 4-week intervals without long-lasting steroid-sparing control of anterior chamber inflammation or CME. At this point, infliximab was changed to weekly 162 mg subcutaneous tocilizumab (Actemra, Genentech). After two doses, CME completely resolved, and BCVA improved from 20/40 to 20/25. The patient was tapered off topical steroids and continues on methotrexate and tocilizumab dual therapy, with excellent control of anterior uveitis and CME.
Discussion
CME is a known complication and cause of vision loss in patients with chronic noninfectious uveitis,1 often persisting after uveitis is seemingly controlled.2 CME occurs due to disruption of the blood-retinal barrier, leading to intracellular and extracellular fluid accumulation within the retina.3 Treatment of uveitis and associated CME involves local and/or systemic corticosteroids and can require immunosuppressive agents, such as antimetabolites and biologics.4,5
FAST FACTS ON IL-6
- Has been found in significant concentrations in the aqueous and vitreous of patients with chronic uveitis
- Induces an excess of VEGF, resulting in angiogenesis and vascular permeability
- Is implicated in the development of CME
Chronic uveitis and associated CME are thought to be mediated in part by interleukin-6 (IL-6), as significantly elevated concentrations of IL-6 have been found in the aqueous and vitreous of patients with chronic uveitis.6 In the eye, IL-6 induces an excess of vascular endothelial growth factor, resulting in angiogenesis and vascular permeability, and, thus, is implicated in the development of CME.7
Tocilizumab is a humanized monoclonal antibody that inhibits IL-6 receptors. Off-label use of tocilizumab has shown utility in patients with uveitis and uveitis-related macular edema.8 Preliminary 6-month results of the ongoing STOP-Uveitis study investigating the safety, tolerability, and efficacy of tocilizumab in patients with noninfectious uveitis have demonstrated that tocilizumab is effective in improving noninfectious uveitis and its complications.9 Anti-IL-6 therapy can be considered for refractory noninfectious uveitis and associated CME.
1. Lardenoye C, van Kooij B, Rothova A. Impact of macular edema on visual acuity in uveitis. Ophthalmology. 2006;113(8):1446-1449.
2. Levin M, pistilli M, Daniel E, et al. Incidence of visual improvement in uveitis cases with visual impairment caused by macular edema. Ophthalmology. 2014;121(2):588-595.
3. Munk M, Kiss C, Steiner I, et al. Systematic correlation of morphologic alterations and retinal function in eyes with uveitis-associated cystoid macular edema during development, resolution and relapse. Br J Ophthalmol. 2013;97:1289-1296.
4. Ossewaarde-van N, Rothova A. Clinical review: update on treatment of inflammatory macular edema. Ocul Immunol Inflamm. 2011;19(1):75-83.
5. Markomichelakis N, Theodossiadis P, Pantelia E, et al. Infliximab for chronic cystoid macular edema associated with uveitis. Am J Ophthalmol. 2004;138(4):648-650.
6. Perez V, Papaliodis G, Chu D, et al. Elevated levels of interleukin 6 in the vitreous fluid of patients with pars planitis and posterior uveitis: the Massachusetts Eye & Ear experience and review of previous studies. Ocul Immunol Inflamm. 2004;12(3):193-201.
7. Mesquida M, Molins B, Llorenc V, et al. Targeting interleukin-6 in autoimmune uveitis. Autoimmun Rev. 2017;16(10):1079-1089.
8. Adan A, Mesquida M, Llorenc V, et al. Tocilizumab treatment for refractory uveitis-related cystoid macular edema. Graefes Arch Clin Exp Ophthalmol. 251(11):2627-2632.
9. Sepah Y, Sadiq M, Chu D, et al. Primary (month-6) outcomes of the STOP-Uveitis study: evaluating the safety, tolerability, and efficacy of tocilizumab in patients with noninfectious uveitis. Am J Ophthalmol. 2017;183:71-80.






