Editorially independent content supported with advertising from


The management of a glaucoma patient who has manifestations of a progressive neuropathy (or whose disease is perceived as not being controlled) is complex and multifactorial. The first question is whether the IOP is on target. If not, the next step is to determine the reason for the lack of control.
LOSS OF EFFICACY
If the patient’s medications are no longer effective, I consider adding to or altering the therapeutic regimen.
In my practice, if a patient is using two medications, he or she is usually administering a prostaglandin analogue (PGA) in the evening and timolol in the morning. If I were to add to this drop regimen, I would nearly always switch the patient to a fixed combination that includes a β-blocker, which is usually an easy transition, because the patient is already on timolol. If he or she cannot tolerate a β-blocker, then I consider a fixed combination of brimonidine and brinzolamide.
An additional option that is very reasonable, especially if I need to lower the IOP by just a few millimeters of mercury, would be to consider laser trabeculoplasty (LTP). This procedure has been consistently shown to be relatively safe, effective, and well tolerated.1 The obvious benefit of LTP is that no agent is added to the patient’s medication regimen. Minimizing a patient’s drop burden has been shown to decrease ocular surface toxicity, minimize the cost of medication, and increase compliance and adherence.2
PROBLEMS WITH ADHERENCE
It is necessary to consider whether patients are using their prescribed medications. It is also important to evaluate both the patient and his or her lifestyle to determine whether topical drops are a realistic long-term solution. Several studies have shown that doctors are usually poor judges of whether a patient is likely to adhere to prescribed medical therapy. That said, I always attempt to tailor patients’ therapeutic regimens to their lifestyle.
For example, the situation of an 80-year-old retired patient with a family member or nurse who assists with drops is very different than that of a 40-year-old patient who travels routinely for work and is always on the go. In the former case, if the IOP is not controlled, I am more likely to add another medication or to switch from a β-blocker to a fixed-combined medication (dorzolamide-timolol or brimonidine-timolol). In the latter situation, I will likely explore surgical options that minimize or eliminate drops.
MICROINVASIVE GLAUCOMA SURGERY
Options
The growing number of options for microinvasive glaucoma surgery (MIGS) has changed my approach to a patient on a few medications whose IOP is no longer controlled. Given recent advances in technique, technology, and safety, I am more likely today to intervene surgically in cases of uncontrolled disease. If I have a patient with definitive visual field loss or significant nerve fiber layer loss, I will seriously consider an isolated angle-based procedure such as GATT (Figure 1) or a limited ab interno trabeculotomy using the Kahook Dual Blade (KDB; New World Medical; Figure 2). Trabectome surgery (NeoMedix) is another option. These surgeries have been shown to be relatively effective in lowering IOP and decreasing drop burden; moreover, they are relatively well tolerated and safe.3-5 An additional consideration is that GATT and the KDB and Trabectome procedures can be performed with or without cataract surgery.
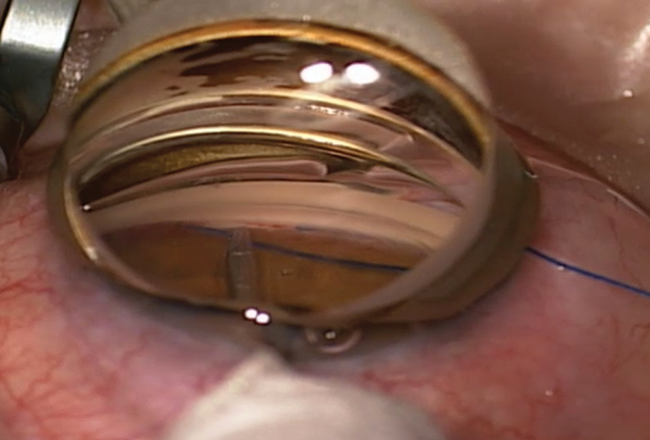
Figure 1. Gonioscopy-assisted transluminal trabeculotomy (GATT) performed with a 5–0 Prolene suture (Ethicon) and a Swan-Jacobs gonioprism. The suture passes nearly 360º around the angle.

Figure 2. Ab interno trabeculotomy performed with a KDB. A small segment of Schlemm canal has been opened (area immediately to the right of the blade), and the instrument is incising the trabecular tissue (area to the left of the blade).
The lens status of a patient plays a role in my decision on adjusting medical therapy, performing LTP, or jumping to a MIGS procedure. The absence of a cataract that is visually bothersome or contributing to outflow problems (inducing angle-closure glaucoma) can make it difficult to justify the risks of incisional surgery. The presence of such a cataract, however, prompts me to consider extraction and one of the various MIGS procedures that can be safely and effectively performed concurrently, including the CyPass Micro-Stent (Alcon; Figure 3), GATT, iStent Trabecular Micro-Bypass Stent (Glaukos; Figure 4), KDB, and Xen Glaucoma Treatment System (Allergan).
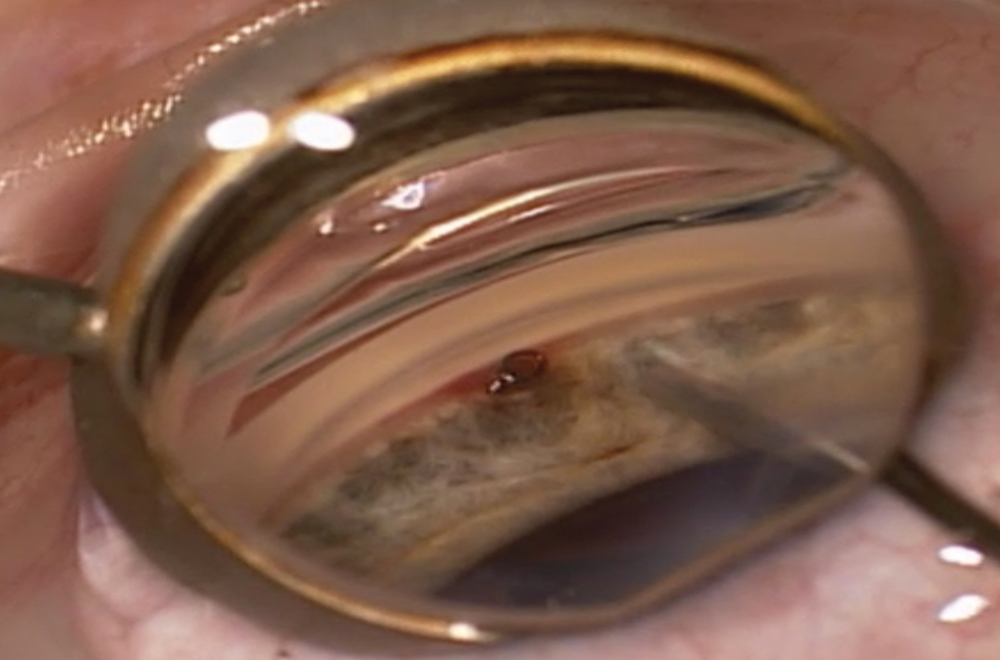
Figure 3. Intraoperative gonioscopy shows the CyPass in the supraciliary space. The implant is ideally positioned, away from the cornea and iris with a collar and one ring visible.
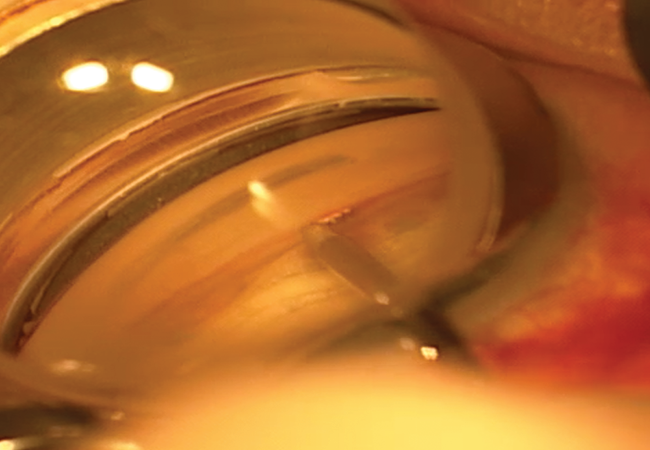
Figure 4. Intraoperative gonioscopy during implantation of the iStent. Note that the surgeon is placing the device through the trabecular meshwork, which can be appreciated given the moderate degree of pigment.
My Approach
Typically, especially in my patients with mild glaucoma, I will perform a surgery that enhances inherent outflow (GATT, iStent, or KDB) before considering creating a new outflow system (Xen). I have yet to come up with clearly defined indications for when I will consider a CyPass versus an iStent. Based on my experience with these devices, the iStent may produce modest IOP lowering and allow me to stop a single medication or to maintain the patient on the same number of medications. I feel as if the CyPass may have a slightly greater IOP-lowering effect than the iStent, but especially in eyes with thick sclera, I have had cases of persistent choroidal fluid and a myopic shift that lasted longer than 3 months.
If the patient requires a significant IOP reduction, then I generally favor surgery with the KDB or GATT. Their disadvantage in these situations (compared with the iStent and CyPass) is that, sometimes, the time to visual and physical activity recovery is longer. In most cases, I find that patients who receive the iStent and CyPass experience a rapid visual recovery and can return to regular activity in about the same amount of time as after isolated cataract surgery. A hyphema (usually microhyphema) is visible after GATT or KDB surgery and usually resolves after 1 to 2 weeks.
When a MIGS-type procedure does not adequately control the IOP, I consider escalating medical therapy or performing a more traditional glaucoma surgery (ie, trabeculectomy or tube shunt), depending on the patient and the stage of disease.
CONCLUSION
It is an exciting time for ophthalmologists and their patients afflicted with glaucoma. Numerous MIGS procedures can be safely performed in isolation and in combination with cataract surgery to help manage uncontrolled disease. In patients whose glaucoma is not controlled on two medications, it is important to consider LTP or the introduction of a fixed-combination medication to further reduce IOP without increasing the patient’s drop burden. Now, more than ever, ophthalmologists are able to tailor the treatment plan to the specific patient’s age, lifestyle, and stage of disease.
1. Zhou Y, Aref AA. A review of selective laser trabeculoplasty: recent findings and current perspectives. Ophthalmol Ther. 2017;6(1):19-32.
2. Robin A, Grover DS. Compliance and adherence in glaucoma management. Indian J Ophthalmol. 2011;59(suppl):S93-96.
3. Grover DS, Godfrey DG, Smith O, et al. Outcomes of gonioscopy-assisted transluminal trabeculotomy (GATT) in eyes with prior incisions glaucoma surgery. J Glaucoma. 2017;26(1):41-45.
4. Grover DS, Godfrey DG, Smith O, et al. Gonioscopy-assisted transluminal trabeculotomy, ab interno trabeculotomy: technique report and preliminary results. Ophthalmology. 2014;121(4):855-861.
5. Khouri AS, Wong SH. Ab interno trabeculectomy with a dual blade: surgical technique for childhood glaucoma. J Glaucoma. 2017;26(8):749-751.
