When opening a traditional glaucoma textbook, it is unlikely to find a chapter titled, “Dry Eye Disease in the Glaucoma Patient,” or something in that neighborhood—perhaps as unlikely as finding a chapter on microinvasive glaucoma surgery. As new technology, surgical techniques, and approaches in management emerge, we first hear about them through trade journals, meetings, and informal exchanges among colleagues.
In this column, I would like to discuss a topic that is often overlooked in a busy comprehensive or glaucoma clinic: dry eye disease (DED). DED, particularly the evaporative type, has been the topic “du jour” in recent years, especially as we have experienced and proven to ourselves how a poor ocular surface can destroy all of the hard work performed in refractive surgery. An undiagnosed, untreated dysfunctional ocular surface can lead to a very unhappy cataract surgery patient, and this can be a source of considerable frustration for doctor and patient alike. It is of equal importance to examine, diagnose, and treat DED in our glaucoma patients as well. Not only is it the right thing to do, but it will also go miles when it comes to creating or enhancing empathy and rapport with our patients.
Every ophthalmologist who treats glaucoma is familiar with the challenges involved in recommending a treatment for an asymptomatic disease, especially when the treatment may negatively affect the patient’s quality of life. A patient could potentially have to jump so many hurdles to be compliant that I am sometimes surprised by how many of them actually do follow instructions. Patients must remember to use the drop; manage to place the drop in (not around) the eye; tolerate potential pain, burning, or blurry vision from or exacerbated by the drops; and, in many cases, pay a good amount of money for their medications. All of these factors—cost, forgetfulness, hand-eye coordination, and poor tolerance to uncomfortable topical medication side effects—are well-known factors that contribute to the low compliance seen in glaucoma treatment. Because of this, anything we ophthalmologists can do to increase a patient’s compliance with treatment becomes highly important. Treating DED is one of those things.
In my clinic, there are days when I am left with the feeling that almost everyone has a bad ocular surface. How symptomatic patients are and how likely they are to spontaneously bring up their symptoms varies tremendously from patient to patient and does not always correlate with the exam findings. Objectively speaking, studies have found ocular surface disease symptoms to be present in about 50% of patients with glaucoma.
Practicing as a glaucoma specialist has increased my interest in staying up to date with DED and its treatment. Improving the ocular surface baseline before starting a glaucoma topical treatment regimen or addressing it in a chronic patient with years of discomfort with a topical treatment can make a big difference in a patient’s life. Glaucoma drops are likely to be better tolerated in patients who do not have a dysfunctional ocular surface. Also, all of our glaucoma tests are dependent on a healthy, smooth ocular surface for better visual acuity in visual field testing and for the best test reliability on imaging structural tests.
PEARLS FOR MANAGING DED
Diagnosing DED or Ocular Surface Disease
Listen to patients and address their symptoms. Don’t let the discussion of a patient’s dry eye symptoms get lost or forgotten in a lengthy conversation about glaucoma and the therapeutic alternatives or steps to be taken for its management. Just like in glaucoma, there can be many misconceptions regarding dry eyes that deserve to be clarified for better participation of patients in their own care. Understandably, many patients are bewildered when learning that their constant tearing could be secondary to dry eyes. Also, many won’t realize that dry eyes, sometimes perceived as a simple, less important problem, could lead to blurry vision, and this is one of the most common and disruptive symptoms present in patients with a dysfunctional tear film.
Perform a thorough eye exam. The eyelid position and margin, status of the meibomian glands (evaluating if clogged or not and the characteristics of secretions), as well as the corneal epithelium should be examined routinely.Determine what type of DED is predominant. My Basic and Clinical Science Course cornea book from residency (2003) states that the most common type of dry eyes is that of aqueous tear deficiency, as opposed to newer evidence indicating a heavier presence of the evaporative type, most commonly caused by meibomian gland disease. As more objective and quantitative ways to study the ocular surface emerge, we will continue to learn more about the tear film and its quality.
Treating Ocular Surface Disease in the Glaucoma Patient
Lid hygiene with warm compresses and lid scrubs, environmental and nutritional factors. Warm compresses can be very tedious and impractical to incorporate into daily life, but they can help and are still a worthwhile suggestion. Cleaning along the eyelid margins with a gentle cleanser, such as a diluted baby shampoo, or marketed lid scrubs, such as OcuSoft (OcuSoft Inc.) or steriLids (TheraTears), seems to be very helpful for some patients. A discussion about environmental factors is important, recommending patients avoid aggravating factors such as air conditioning and smoke and consider humidifiers and goggles that may bring relief. Also, recommending that patients eat foods rich in omega-3 or take omega-3 and/or fish oil supplements can help patients improve the quality of their tear film.
Artificial tears, gels, and ointments. I think it is best to recommend one or two types of products and then leave it to the patient to choose which to use on their own. As more is learned about the tear film, more specific artificial tears can be recommended based on both the severity and type of dry eye. Many options are available in both preserved and nonpreserved modalities.
Punctal plugs. I sometimes feel punctal plugs are overused and, on many occasions, placed in patients who might benefit from an improvement of the quality of the tear film before getting plugs; their symptoms could actually be worsened by premature placement of plugs and retention of all inflammatory mediators already present in the tear film.
Topical cyclosporine (Restasis; Allergan, Inc.). I tend to incorporate Restasis early on, especially as new evidence seems to support its earlier use.
Steroid drops along with Restasis. I use Alrex (Bausch + Lomb) or Lotemax (Bausch + Lomb) for a limited time, usually about a month with adequate IOP monitoring and very clear instructions and warnings about the possibility of a steroid response with unlimited chronic use.
Tetracyclines. Topical azithromycin drops (AzaSite; Akorn, Inc.) and oral doxycycline of20 mg to 50 mg once or twice daily are commonly prescribed for DED as well. When prescribing doxycycline, it becomes important to provide some information to patients such as, take with food but not with high-calcium food, stay away from the sun, and do not use if there is a diagnosis of esophagitis.
Selective laser trabeculoplasty (SLT). Dry eye or not, I present SLT to all eligible patients as a first-line treatment. I present this option along with the option to use a glaucoma drop. After I’m done explaining the pros and cons of each alternative, I would say that more than half of my patients choose SLT over drops, especially those with dry eyes.
If the severity or threat of glaucomatous damage allows, I try to improve the ocular surface before starting any glaucoma treatment in a newly diagnosed glaucoma patient. Some cornea and glaucoma specialists might feel comfortable pursuing a drug holiday in patients undergoing glaucoma topical treatment who have a poor ocular surface. Notifying the specialist who is treating the patient’s glaucoma is a must to avoid any unwanted advancement of glaucomatous damage.
Topical medical treatment. With mounting evidence of the negative effects of preservatives on the ocular surface, decreasing a patient’s exposure to preservatives, especially to benzalkonium chloride (BAK), is reasonable when treating a chronic disease such as glaucoma that more often than not requires multitherapy. During the past few years, there has been an increase in the number of available preservative-free and non-BAK preserved options, as listed below.
Nonpreserved Glaucoma Topical Medications Available in the United States
•Preservative-free timolol maleate (Timoptic in Ocudose; Aton Pharma)
•Preservative-free tafluprost (Zioptan; Merck & Co., Inc.)
•Preservative free dorzolamide hydrochloride-timolol maleate (Cosopt PF; Akorn, Inc.)
Non-BAK Preserved Glaucoma Drops
•Brimonidine purite (Alphagan P; Allergan, Inc.)
•Travoprost with sofZia (Travatan Z; Alcon Laboratories, Inc.)
New treatment modalities. The LipiFlow Thermal Pulsation System (TearScience) is the therapeutic arm of LipiView and is a 12-minute treatment that warms up and massages the meibomian glands, improving their function and the lipid layer of the tear film. It seems to be beneficial in some patients with meibomian gland dysfunction by improving symptoms for several months. The treatment is applied using a single-use eyepiece that treats the meibomian glands in both the upper and lower lids simultaneously. Patients usually describe feeling a warming sensation across the eyelids during treatment.
Intense pulsed light (IPL) therapy is a light-based treatment that has been used for a long time in dermatology to treat rosacea and other vascular facial lesions. By applying specific wavelengths of light to target abnormal blood vessels or telangiectatic vessels in the eyelids, causing ablation, IPL can improve inflammation and meibomian gland dysfunction. Many patients with evaporative DED have associated ocular rosacea and could benefit from this relatively new application of IPL. Just like LipiFlow, this is a new treatment modality worth exploring further, as it may provide long-term improvement of symptoms. The treatment takes about 10 to 15 minutes. A protective eye shield is placed over the patient’s eyes.
Other. There are some patients for whom glaucoma surgery becomes instrumental not only to halt glaucoma progression but also to improve their ocular surface. This can be the case in patients with increased sensitivity to preservatives and chemicals from the several drops they may be using.
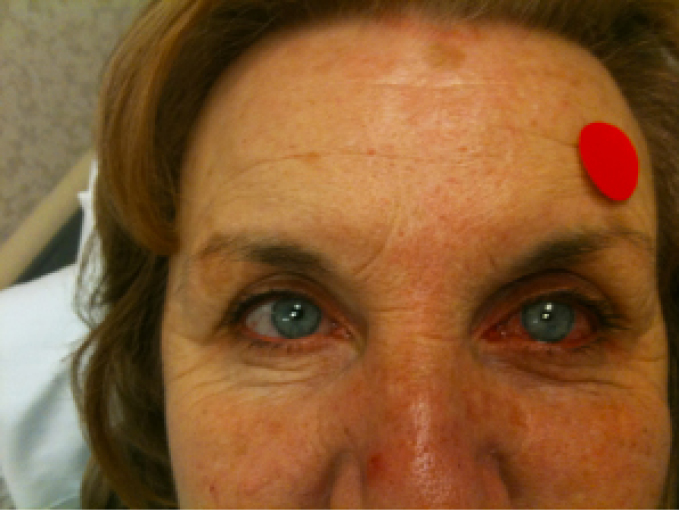
This is a patient with advanced and uncontrolled glaucoma who had Ex-Press Mini Glaucoma Shunts placed. You can appreciate how quiet the right eye is in comparison to the left eye. The right eye underwent Ex-Press Mini Glaucoma Shunt implantation about 2 weeks prior to this photo and had achieved the target IOP without the use of drops, while the left eye was still on three to four drops. This photo was taken the day of (and prior to) Ex-Press Mini Glaucoma Shunt implantation in her left eye.
CONCLUSION
Diagnosing and treating DED is more than rewarding when it comes to patients with glaucoma. It can improve their overall vision, their eye comfort, their performance on diagnostic glaucoma tests, their tolerance of glaucoma topical treatments, their compliance with glaucoma medications, and their overall trust in their ophthalmologist.
Given the chronic nature of glaucoma and the common need to use multiple topical medications to control IOP, it is vital that we take into account the presence of DED to better individualize treatment. We should make use of options available to us to decrease exposure to preservatives, as their toxicity is dose-dependent. In addition, ocular toxicity can occur secondary to the medications themselves. The medical regimen should be kept as minimal as possible, ensuring that all medications provide a justifiable IOP-lowering effect, adding medications one by one when possible, and making use of combination medications when feasible.



