For cataract surgeons, the achievement of optimal outcomes is a constant pursuit. To facilitate this undertaking, numerous technologies and techniques have emerged, most of which come into play during the preoperative and intraoperative periods. Surgeons work diligently to measure the refraction of the affected eye, select the best IOL, set appropriate patient expectations, and confirm the accuracy of their measurements. All of these steps are taken in an attempt to ensure the patient’s postoperative satisfaction and, thus, prevent the need for spectacles or lens explantation and/or exchange.
Despite surgeons’ efforts to promote patient satisfaction, it is impossible, given the vagaries of surgery and human physiology, to guarantee a perfect outcome that forever stands the test of time. The literature suggests that 67.5% of patients with co-pathology and 49.2% patients without co-pathology do not have 20/20 visual acuity after cataract surgery. The combination of surgical complications and refractive issues leave many patient with residual postoperative errors.1 In addition, when premium lenses are employed, patient expectations and desires are frequently not in sync with the limitations of the existing technology. The patient may be disappointed by the halos and glare that can accompany premium lenses in some cases. All of these factors have the potential to contribute to a patient’s dissatisfaction with his or her cataract surgery outcome.
What if, rather than (1) removing the implanted IOL, (2) prescribing glasses, or (3) counseling the patient, there was a way to modify the lens in vivo to the patient’s and physician’s satisfaction? That is precisely what the Perfect Lens technology is able to do.2
MISSION AND TECHNOLOGY
The Perfect Lens technology was conceived in 2009 with the goal of determining a way to modify an acrylic lens, either in vitro or in vivo, using a femtosecond laser and the two-photon process. The company is not a lens manufacturer and does not manufacture an IOL. The Perfect Lens technology can be used to alter the refractive index of acrylic material and thus alter the refractive characteristics of an IOL in vitro or in vivo. This technology works with standard commercially available hydrophobic and hydrophilic IOLs.
With the Perfect Lens approach, in vivo modification is achieved by creating a lens within an existing IOL using the light from a femtosecond laser. The lens created within the implanted lens is between 4.0 and 4.5 mm in diameter but only 50 to 100 μm in depth, thus fitting well into the existing IOL. The lens within the lens is created by altering the hydrophilicity of the IOL in certain designated areas. This alteration of hydrophilicity and the subsequent absorption of water by the material alters the refractive index of the implanted lens and thus its refractive characteristics. The shape, size, and external features of the implanted lens are unaffected. The changes created by the Perfect Lens technology alter the IOL and enable the surgeon to create the patient’s desired vision in a simple postoperative in-office procedure.

Figure 1 | The Perfector.
The machine housing the femtosecond laser—the Perfector (Figure 1)—is mobile and uses a proprietary docking attachment that is designed to utilize less pressure than existing capsulorhexis machines. This low-pressure design increases patient comfort.
Once the physician has examined the patient and identified the refractive error of the implanted IOL, he or she then enters the details of the implanted lens and the desired change to the IOL into the computer console of the Perfector. The patient is attached to the Perfector using the patient attachment. An optical coherence tomography (OCT) system within the Perfector is used to locate the lens and identify the area within the IOL where the new lens will be constructed. The computer console on the Perfector shows the physician exactly where the laser is focused within the lens. Once the OCT has accurately focused the laser, the physician initiates the laser. The creation of the lens within the IOL takes between 20 and 40 seconds.
THE UNMET NEED
For most physicians, the primary barrier to adoption of multifocal IOLs is the potential risk of glare and halos, with the unhappy patient requiring counseling or, in extreme conditions, explantation. The Perfector resolves this issue by offering the ability to simply remove multifocality from a multifocal lens, thus eliminating the offending halos and glare.
Another aspect of the Perfect Lens technology is its ability to add premium functions to an implanted IOL. A patient previously implanted with a monofocal IOL can add multifocality if he or she wants to try life without glasses. If an IOL was implanted in the past and the patient’s vision has changed, the physician can adjust the IOL to fit the new parameters. The ability to alter the lens in vivo helps physicians meet patients’ needs as they evolve throughout life.
PROOF-OF-CONCEPT STUDIES
Perfect Lens has conducted numerous in vitro studies and has consistently demonstrated the ability to alter the IOL diopter to within 0.10 D of the desired goal, with a diminution of modular transfer function of less than 0.1 or a final modular transfer function above 0.5 for the 100 lp/mm measurement. Perfect has observed no significant forward scatter induced within the lens, no detectable leachables, and no measureable chromatic aberration. The current limit to the adjustment of an implanted IOL is approximately 4.00 D. The adjustment is stable, safe, and effective for the patient.
ACTUAL RESULTS
Different lens types and lens materials have been tested extensively. One material was used to test a large variety of lens types and shaping options, including asphericity, toricity, diffractive multifocal lenses, refractive multifocal lenses, multifocality cancellation, multiple treatments, and cancellation of the shaped lenses. All were done successfully.
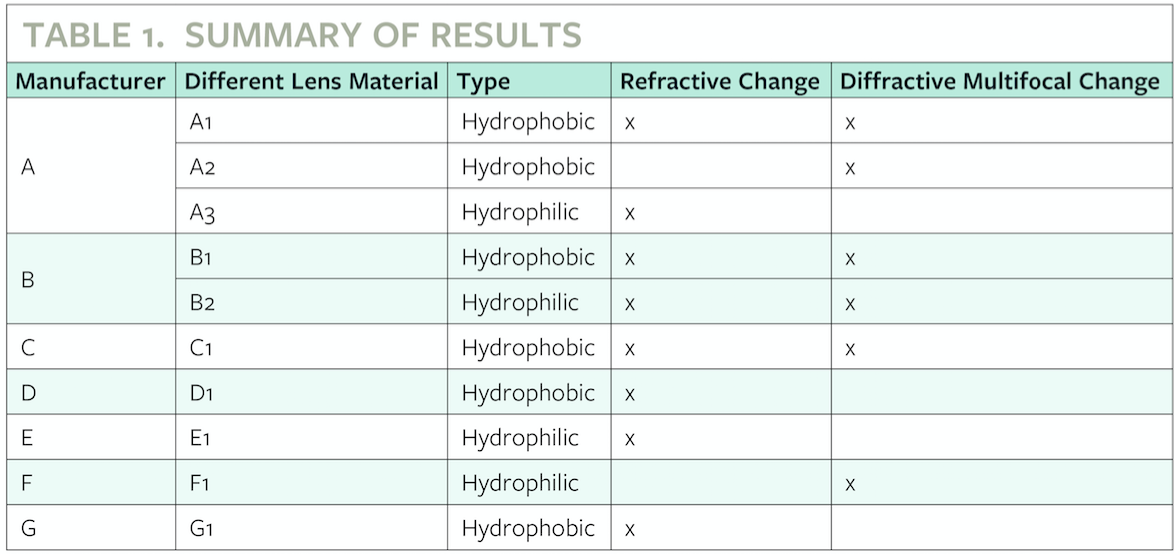
Table 1, shown above, displays the laboratory results of the Perfect Lens technology on a variety of available IOL materials from different manufacturers. Additional outcomes are shown below in Figure 2.
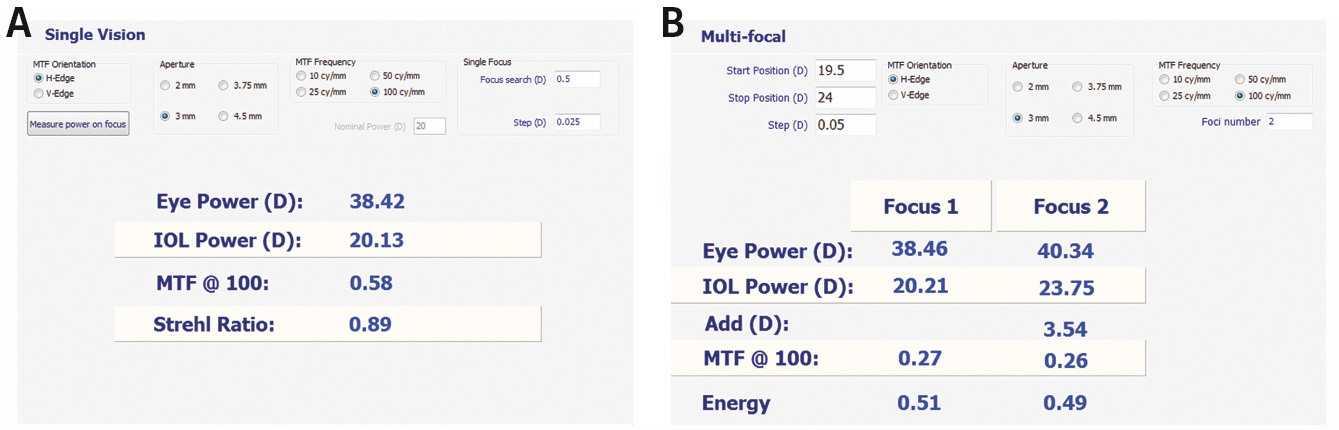
Figure 2 | Results before (A) and after (B) refractive index shape change, showing the creation of multifocality in a monofocal hydrophobic IOL.
FUTURE OUTLOOK
The Perfector has altered the power, asphericity, multifocality, and toricity of hydrophobic and hydrophilic lenses. A study in rabbit eyes is currently under way at Moran Eye Center, and results are expected in the beginning of 2017.
1. Day A C, Donachie P H J, Sparrow J M, Johnston R L. The Royal College of Ophthalmologists’ National Ophthalmology Database study of cataract surgery: report 1, visual outcomes and complications. Eye. 2015;29:552-560
2. Sahler R, Bille JF, Enright S, Chhoeung S, Chan K. Creation of a refractive lens within an existing intraocular lens using a femtosecond laser. J Cataract Refract Surg. 2016;42(8):1207-1215.





