Minimally invasive and powerfully effective. These are words rarely heard together in the context of moderate or severe glaucoma treatment—until now. The Xen Glaucoma Treatment System from Allergan allows us to access a new pathway for aqueous flow from the anterior chamber to the subconjunctival space.
The system includes a preloaded, disposable injector (Figure 1) to insert a 6-mm length, 45-micron lumen diameter gel stent via an ab-interno approach. Aqueous flow controlled through lumen restriction creates a diffuse, low-lying, posterior, ab interno bleb without disturbing Tenon capsule adhesions or low-lying drainage space.
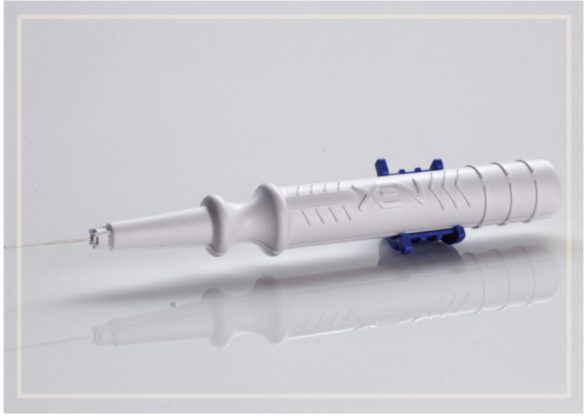
Image courtesy of New World Medical
FIGURE 1. A preloaded, disposable injector is used to insert a 6-mm length, 45-micron lumen diameter gel stent via an ab interno approach.
The gel stent is clinically proven to reduce mean IOP and the number of IOP lowering medications. The pivotal trial established an excellent safety profile with zero intraoperative complications or persistent hypotony.
Glaucoma specialists involved in the development and study of this gel stent share their experience and insight with us.
Arsham Sheybani, MD
True glaucoma procedures, such as tube shunts and trabeculectomy, can be the bane of the glaucoma surgeon’s existence. I have left surgeries where all proceeded perfectly with indications of a good outcome only to see a very different result the next day, particularly when it comes to IOP. The variation between patient outcomes and digression of results occurs because of unpredictable aqueous flow. This new device and its safe, reproducible procedure is a promise of reduced post operative visits and an opportunity to help people maintain good quality of life and remain in the workforce.
With this stent, we are starting to see a progressive revolution and standardization of glaucoma surgery comparable to the evolution of phacoemulsification. This new generation of devices can tackle pressures and bring them lower than we have seen before and make patient recovery more reproducible and less difficult. Although this stent is not the magic bullet to treat all glaucoma disease, it offers us the ability to effectively treat a broader range of patients.
A reproducible procedure behaves predictably. This gel stent helps to take subjectivity out of the equation by giving us access to the subconjunctival pathway, the most tried and true way to lower IOP. Patients who produce normal aqueous will typically experience consistent and regular flow through the device immediately upon placement.
As the Baby Boomer population ages, we can expect an inundation of glaucoma disease requiring surgery. The gel stent is a minimally invasive, repeatable procedure. If a patient responds well the first time, we can retreat those who may begin to require medication again.
Presently indicated for management of glaucoma cases where previous treatments and surgeries have failed, I believe that once safety and efficacy are well established, we will see this stent applied for treatment of milder disease, particularly in the patients who must take multiple medications just to maintain slight to moderately high pressures. Simple angle surgery will not enable this class of patients to get off medication; however, this device offers them that chance.
Iqbal “Ike” K. Ahmed, MD
This gel stent (Figure 2) fills a unique position among MIGS and even in terms of glaucoma surgery. While the device exhibits the characteristics for inclusion on the MIGS spectrum, including an ab interno approach, this device is distinguished by an external filtration mechanism. External filtration allows for more potent IOP lowering by creating a bleb and resulting pressures that approach what we are able to achieve with traditional trabeculectomy. Within my practice, we see patient outcomes with IOPs in the 12- to 14-mm Hg range and a reduction in medication requirements from three or four prescriptions to one or none. Additionally, when the procedure is combined with mitomycin C, it appears to enhance efficacy with only a minimal drop off in its safety profile.1 About 70% of patients experience an uneventful postoperative course. The remaining 30% may require additional post-operative intervention such as a subconjunctival injection, bleb needling or back on a medication.
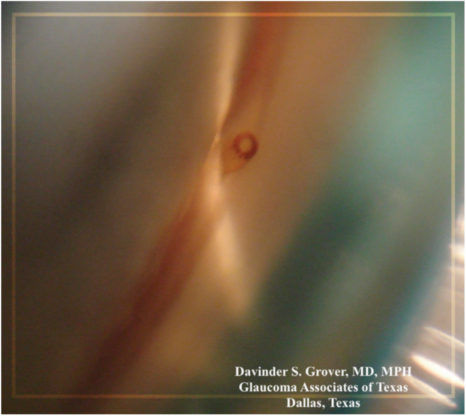
Image courtesy of Davinder S. Grover, MD, MPH
FIGURE 2. Gonioscopic view of the Xen.
The gel stent has broadened our indications and stands to play an important role for patients who fall between the extremes on either end of the glaucoma spectrum. In particular, pre-trabeculectomy patients who are dependent upon three or more medications, but reluctant to proceed to traditional glaucoma surgery due to concerns about durability and safety, can undergo this less invasive procedure in the earlier stages of the disease and achieve comparable results without the complications of traditional trabeculectomy and tube insertion. Patients who require IOP to remain consistently below 10 mm Hg will likely still need a trabeculectomy. On the other end of the spectrum, patients who have ocular hypertension, or who are headed to cataract surgery and have mild glaucoma disease (requiring only one medication) are more likely to benefit from a canal or suprachoroidal internally based procedure.
Richard A. Lewis, MD
This device was developed to be implanted ab interno without opening the conjunctiva, and with mitomycin C and lidocaine injected into the subconjunctival space. However, FDA labeling for mitomycin C is approved only for sponge application, thus requiring the US trial to be conducted differently than the device was intended, and is currently (and successfully) used in Europe and Canada.2
The US pivotal trial of this new gel stent in refractory glaucoma patients showed a reduction in IOP from a mean medicated baseline of 25.1 (+3.7) mm Hg to 15.9 (+5.2) mm Hg at 12 months.3 The FDA trial procedure involved opening the conjunctiva and applying mitomycin C directly with a sponge and suturing the conjunctiva rather than simply injecting the mitomycin. The added level of risk limited trial candidates to those with moderately advanced glaucoma who had failed prior surgical intervention and were uncontrolled with maximum med therapy.
The trial demonstrated a better safety efficacy profile than traditional glaucoma surgery for these patients, and is indicated for use only with moderately advanced glaucoma. Since no other MIGS labeling has this indication, we now have a surgical procedure to offer to patients who fall between the extremes—those with disease too far advanced to respond to trabecular meshwork stent, but not severe enough to warrant the risks associated with tube shunts and trabeculectomy. Most importantly, when we look at the average percentage of IOP reduction in MIGS procedures compared with the Allergan gel stent, the gel stent gets the pressure lower. The requirement of mitomycin C and post-operative management of blebs does slightly offset the advantage of MIGS; however, we already know to expect and manage blebs in refractory glaucoma procedures.
Post surgically, we treat patients with the typical steroids and antibiotics. Hypotony and bleb management are significant concerns associated with filtering surgeries. The design, location, and diameter of the gel stent regulate outflow by reducing flow to prevent hypotony. Although a few trial patients presented with lower pressure, none had clinical signs of hypotony or infection. Trabeculectomy frequently produces a bleb which can be very close to the limbus, ischemic, irritating, and affect visual function. In contrast, the gel stent blebs are more posterior and diffuse, not ischemic, and less irritating. Bleb scarring is usually easily resolved with needling.
1. Errico D, Scrimieri F, Riccardi R, Fedeli R Iarossi G. Trabeculectomy with double low dose of mitomycin C – two years of follow-up. Clin Ophthalmol. 2011;5:1679-1686.
2. Sheybani A, Dick HB, Ahmed II. Early clinical results of a novel ab interno gel stent for the surgical treatment of open-angle Glaucoma. J Glaucoma. 2016;25(7):e691-696.
3. Allergan. XEN Package Insert. For full product information call (800) 678-1605.
Drs. Ahmed, Lewis, and Sheybani serve as consultants for Allergan.


