Presbyopia is the most common inevitable refractive disorder and it is arguably the most frustrating one for patients to deal with. Yet, until recently, there were relatively few options to address the loss of accommodation in precataract patients.
The recent approval by the FDA of the Raindrop Near Vision Inlay (ReVision Optics) significantly alters this paradigm. Simple in design, the Raindrop Inlay is implanted in the stroma of the cornea in the nondominant eye under a femtosecond laser flap, thus changing the shape of the central cornea to provide improvement in near vision (Figure). It is both easy to implant and highly effective, according to Stephen G. Slade, MD, who performed the first implantation of the Raindrop Near Vision Inlay in the United States after FDA approval.
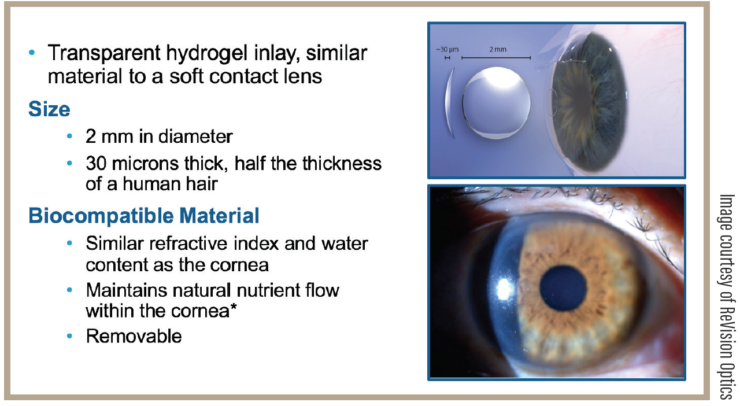
FIGURE. The inlay is made of hydrogel material with water content, and the refractive index is similar to the cornea; 30 μm thick, diameter 2 mm.
In the premarket clinical trials, 92% of patients (336 of 364) achieved uncorrected near visual acuity of 20/40 or better at 24 months of follow-up.1 Although it was not a prespecified endpoint, 76% of patients were 20/25 or better at intermediate distance. It should be noted that this analysis was performed among emmetropic patients from +1.00 D to -0.50 D manifest refractive spherical equivalent.2 Both in the clinical trials and in my own subsequent clinical practice, I have noted that patients with a slight degree of hyperopia achieve the best outcome, as the inlay induces about a diopter of myopic shift. In fact, the FDA approved the device for use in patients with a manifest refractive spherical equivalent of +1.00 D to -0.50 D.2
Relatively few visual or ocular symptoms were noted in the premarket study (the safety profile is somewhat analogous to LASIK). Moreover, distance visual acuity was maintained in over 95% of patients. An important feature for surgeons to keep in mind is that explantation of the inlay is not difficult should that become necessary, with probable reversal of refractive effect.
As far as the procedure itself, any surgeon who is versed in performing LASIK surgery is already well equipped to incorporate the Raindrop into regular patient care. “Several steps of the procedure are identical to LASIK, whereas others are very closely related,” says Dr. Slade, who is in private practice at Slade and Baker Vision in Houston. “For instance, the flap cut is performed at 30% of the corneal depth, which may be slightly deeper than what most surgeons are accustomed to with refractive procedures. Nevertheless, cutting the flap is exactly the same, as is the need to maintain the moisture of the flap during the procedure. Interestingly, the thicker flap created for the inlay may confer a slight benefit. In the cases I have performed, I have noted that the thicker flap tends to flip back and settle more naturally.”
This inlay represents a paradigm shift in treatment of presbyopia and it may require a little bit of a rethink on the part of surgeons, says Dr. Slade. Many LASIK surgeons are accustomed to a “catch and release” model of care, in which effort is exerted to create high surgical volume, but those patients do not necessarily stay with the practice. Offering patients the Raindrop inlay, on the other hand, may be a boom for patient retention. “Because the ideal candidate is someone who is not quite eligible for LASIK but who does not yet need a presbyopia correcting IOL, it fills a currently underserved gap in the continuum of eye care, especially because the inlay does not preclude later cataract surgery when it becomes necessary,” he says. “This ‘bridge’ procedure is perfect to help keep our patients in the practice, ‘catch and keep.’”
1. Raindrop Corneal Inlay Directions For Use.
2. FDA. www.accessdata.fda.gov/cdrh_docs/pdf15/P150034c.pdf. 304-0075 Rev. 4. Accessed April 14, 2017.
Dr. Slade serves as a consultant for Revision Optics.


