With the recent approval by the FDA of the Small Incision Lenticule Extraction procedure (SMILE; Carl Zeiss Meditec), refractive surgeons have a new option in their armamentarium for the correction of myopia. This announcement is poised to reinvigorate the refractive market. For myriad reasons, including concerns over flap-based procedures, economics, and misplaced safety concerns about LASIK, interest in refractive procedures has declined over the past decade, even as refinements in the techniques and technology have made outcomes more predictable and repeatable.
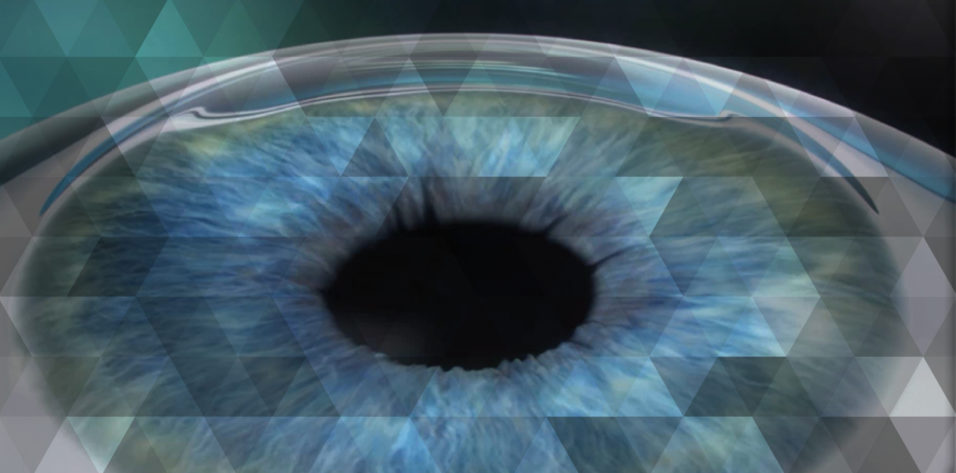
The release of SMILE should really be seen as a win for patients, especially those who might have been considered less than ideal candidates for refractive procedures in the past. Patients with dry eye are an example. With SMILE, which currently can only be performed using the VisuMax Femtosecond Laser (Carl Zeiss Meditec), the size of the corneal incision is considerably smaller than in LASIK and there is evidence that the procedure may sever fewer corneal nerves, both factors that theoretically reduce the risk for dry eye (Figure).1 This could have additional benefit for those with preoperative dry eye, as it may facilitate an easier, more comfortable healing experience.
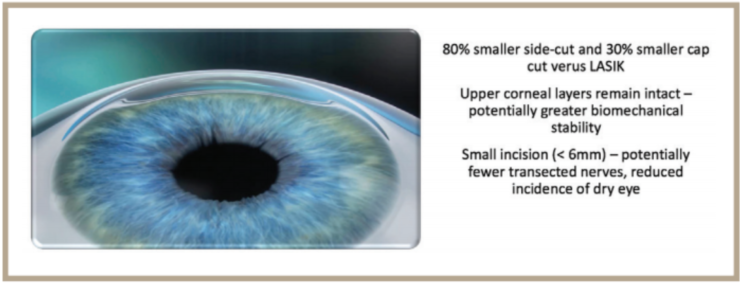
Courtesy of Carl Zeiss Meditec
FIGURE . Characteristics and possible benefits of SMILE.
There has been some discussion that the visual recuperative phase after SMILE may be slightly longer compared with LASIK. “I think some of this may be confusion with an earlier iteration of SMILE called FLEx (Femtosecond Lenticule Extraction), which was associated with slightly protracted time to regain vision,” says John F. Doane, MD, who is in private practice with Discover Vision Centers, Kansas City, Mo., and is clinical assistant professor with the Department of Ophthalmology, Kansas University Medical Center, Kansas City, Kansas.
Another notable feature of SMILE is that because the treatment is not dose independent, outcomes are equally predictable among patients with low or high levels of myopia. As such, the only question this poses is whether correction in low myopes is similar to what is achievable with LASIK, Dr. Doane says.
The clinical study that led to its FDA approval suggests that outcomes are excellent with the SMILE procedure: 327 of 328 treated eyes achieved 20/40 or better vision at 6 months and 88% were 20/20 or better. Issues related to loss of suction and complications, such as dry eye and moderate to severe glare and halo, were noted, with rates comparable to historical numbers after LASIK.2 In addition to these data points, it is also reassuring to know that to date, more than 750,000 SMILE procedures have been performed worldwide since the procedure became commercially available 6 years ago, with clinical outcomes comparable to LASIK.3
SMILE is currently FDA approved for use in the reduction or elimination of myopia in patients with -1.00 D to -8.00D, less than or equal to -0.50 D of cylinder and MRSE of less than or equal to -8.25 D in the eye(s) to be treated and in patients who are aged 22 years or older with documentation of stable manifest refraction over the past year Currently, SMILE is not approved for treatment of astigmatism nor hyperopia. However, a study examining the feasibility of using SMILE to treat myopic astigmatism—is already underway in the United States. Additionally, investigators outside the United States are studying methods for correcting hyperopia with SMILE. “It will be interesting to note what the reaction to SMILE will be in the market. If history is any predictor, novel techniques attend to attract patients’ attention. In fact, LASIK really grew to prominence because of patient demand, and it was refined over the years because patients wanted even better, more predictable, and safer results,” Dr. Doane says. “
1. Denoyer A, Landman E, Trinh L, et al. Dry eye disease after refractive surgery: comparative outcomes of small incision lenticule extraction versus LASIK. Ophthalmology 2015;122(4):669–676.
2. FDA News Release: FDA approves VisuMax Femtosecond Laser to surgically treat nearsightedness. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm520560.htm. Updated September 13, 2016; Accessed May 26, 2017.
3. Comparison of visual ad refractive outcomes following femtrosecond laser assisted LASIK with SMILE in patients with myopia or myopic astigmatism. Journal of Refractive Surgery 2014 Sep, Sri Ganesh, Rishika Gupta.
Dr. Doane is a consultant for Carl Zeiss Meditec.