
Greg Parkhurst, MD, FACS
The Refractive Surgery Alliance
The Refractive Surgery Alliance (RSA) is remodeling the future of vision correction surgery in the United States to the point that annual US refractive surgery volume will ultimately be at least five times the 630,000 procedures being performed annually today. Techniques have been perfected, and technologies are now widely available and advantageous for the large majority of patients with refractive disorders. The antiquated fallacy that refractive surgery is a luxury item, only for those who can afford it and willing to take risks with their eyes, is now being completely snuffed out. Refractive surgery practices of RSA members are already consistently seeing double-digit growth month after month.
“I believe that in the next century, history will look back at our time and recognize that the emergence of refractive surgery marked the first time in human history when a debilitating congenital defect was corrected on a mass scale. The impact of refractive surgery on human function, productivity, and quality of life is immeasurable.” –Guy M. Kezirian, MD, FACS
So, what makes the RSA the game changer that it is? At its core, the RSA is about:
• Promotion of the fact that independent vision improves the quality of life via performance, wellness, and beauty;
• A committed belief that every qualified candidate should have refractive surgery;
• Directing all outward messaging to promote public confidence in
the industry instead of individual surgeons or particular technology
self-promotion;
• Quality outcomes through critical patient selection; and
• Providing surgeons access to sophisticated refractive surgery training programs so that ophthalmologists performing refractive surgery are well qualified to perform the full range of corneal and lenticular procedures.
All of this can be accomplished, and at the same time, save patients with ametropia a significant amount of money.
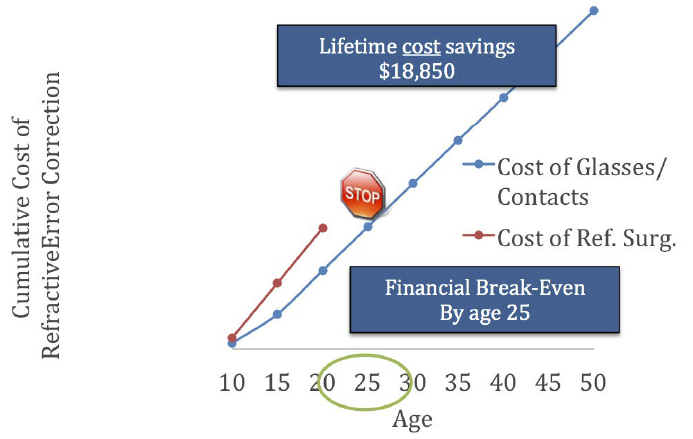
Refractive surgery is positioned to become the next well-recognized “rite of passage” done at the point of ocular maturity, similar to dental braces for children with abnormal teeth structure. Consumers see the obvious performance and cost advantages of permanent vision correction procedures versus the recurring cost and hassle of using prosthetic devices like glasses and contact lenses that only temporarily mask the underlying disease.
US refractive surgery volume is currently well below capacity, partly due to failure in ophthalmic physician leadership, and the RSA is doing something about it. Its mission is to promote the benefits and safe delivery of refractive surgery to every American who wants it and to position US refractive surgery for widespread sustainable growth.
The US market is primed for significant expansion via the RSA’s five pillars of growth:
1. Expanding the education of refractive surgeons to include business administration and entrepreneurial skills via the RSA’s soon-to-be-formed Refractive Surgery Fellowship Network;
2. Changing the capital equipment model and “click per procedure”;
3. Addressing past early-technology unhappy outcomes;
4. Capturing the procedure supply chain at the most advantageous age for the patient; and
5. A parity financing model compared with monthly contact lens expense.
The RSA has well-defined values and specific goals of growing the industry for all patients and refractive surgeons to benefit. Physician colleagues, industry, and patients are invited to join the growing momentum of expanding the industry well beyond its earlier heyday. The RSA is achieving its initiatives via integrity and expertise while avoiding bias and industry-driven positions. In the RSA, ideas are shared and collaboration is encouraged via an overt entrepreneurial spirit of inclusivity.
Greg Parkhurst, MD, FACS, is a cataract and LASIK surgeon at Parkhurst NuVision in San Antonio, Texas. Dr. Parkhurst is the President-Elect of the Refractive Surgery Alliance. He may be reached at:gregory.parkhurst@gmail.com; https://www.sanantonio-lasik.com.

The RSA held its Inaugural Meeting on April 25, 2014 in Boston. From left: Matthias Maus, MD; Wolfgang Riha, MD; Jennifer Loh, MD; Paul Mann, MD; Aaron Waite, MD; Guy Kezirian, MD; Greg Parkhurst, MD; Matthew Hammond, MD; Asim Piracha, MD; Lance Kugler, MD (President); and Jimmy Lee, MD. Not pictured: John Berdahl, MD; Jason Brinton, MD; Dan Durrie, MD; Robert Lin, MD; Richard Lindstrom, MD; Carlos Manrique, MD; Jamie Monroe, MD; Carlos Montoya, MD; Julian Procope, MD; Vance Thompson, MD; Tom Tooma, MD; Steven Vold, MD; David Whiting, MD; Bill Wiley, MD; Helen Wu, MD; and Elizabeth Yeu, MD.
Click here to learn more about the Refractive Surgery Alliance or to join.

V. Nicholas Batra, MD
Trulign Toric IOL
The Trulign Toric posterior chamber IOL (Bausch + Lomb) is intended for primary implantation in the capsular bag for the visual correction of aphakia and postoperative refractive astigmatism secondary to removal of a cataractous lens in adult patients with or without presbyopia; these patient desire reduction of residual refractive cylinder with increased spectacle independence and improved uncorrected near, intermediate, and distance vision.
We utilize the Trulign Toric lens in clinical practice to provide astigmatism correction to patients who have both astigmatism and presbyopia. It is currently the only FDA-approved lens in the United States that has an indication to correct both of these disorders. The ideal patient is an astigmat who performs a variety of activities that require a range of vision. We typically will use the lens when patients have more than 0.75 D of astigmatism. The Trulign Toric IOL has demonstrated its ability to enable patients to work on a computer, drive, go shopping, participate in sports, attend social gatherings, do work around the house, walk, and watch television.1,2
The Trulign provides excellent astigmatism correction when compared with other monofocal toric lenses. It provides excellent visual acuity at intermediate and distance,2 with 97.8% of eyes achieving intermediate UCVA ≥ 20/40 and 97.8% of eyes achieving distance UCVA ≥ 20/40. The Trulign has outstanding rotational stability,2,3 with rotation ≤5o in 96.1% of eyes.2 It provides effective cylinder correction,2 with residual cylinder ≤1.0 D in 95.5% of eyes.2 In addition, 99.2% of patients implanted with the Trulign Toric reported no visual disturbances.2

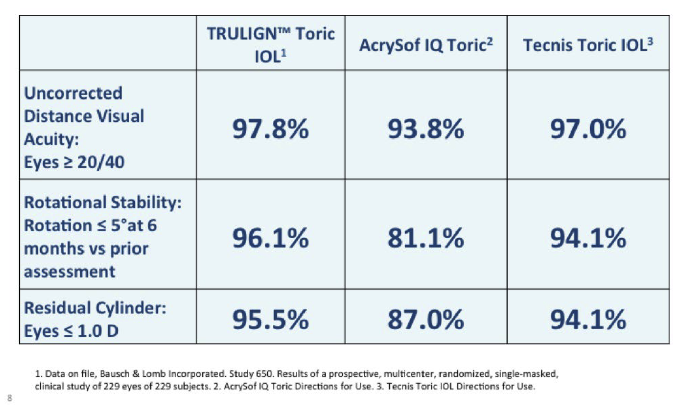
In summary, the Trulign is a game changer because it is the only lens available to correct astigmatism and presbyopia in one step. Previously, many of these patients would have required two procedures—LASIK and presbyopia-correcting IOL implantation—to address their visual needs. This lens allows us to correct both conditions simply and easily, as shown in the video below.
V. Nicholas Batra, MD, is the Medical Director of the Batra Vision Medical Group in San Leandro, California. Dr. Batra states that he is a consultant to Bausch + Lomb. He may be reached at drbatra@batravision.com.
1. Data on file, Bausch + Lomb Incorporated. Study 650. Results of a prospective, multicenter, randomized, single-masked clinical study of 229 eyes of 229 subjects.
2. AcrySof IQ Torc Directions for Use.
3. Tecnis Toric IOL Directions for Use.

Cathleen M. McCabe, MD
Ora With VerifEye
Alcon’s release of the Cataract Refractive Suite and recent acquisition of the Ora System with VerifEye by WaveTec Vision is a real game changer. The integration of preoperative image and data collection with surgical planning, femtosecond laser treatment, intraoperative active aberrometry for plan refinement, and intraoperative incision and lens centration guidance is revolutionizing our quest to improve cataract surgery outcomes. With these technological improvements, we can approach the level of precision seen with refractive surgery procedures and meet the expectations of our patients.
Several new technologies have recently been released or are in development in order to improve visual outcomes following cataract surgery. The IOLMaster (Carl Zeiss Meditec) and Lenstar (Haag-Streit) provide precise preoperative keratometry and axial length measurements. More modern lens formulae (Olsen and Barrett Toric and Universal II) with refinements for long axial length (Wang-Koch adjustment) or posterior corneal curvature (Baylor table) have improved IOL calculations. The Cassini (i-Optics) is a new keratometer and image-capture device based on color LED technology that can be combined with TrueVision heads-up viewing systems for guidance during lens placement and astigmatism management. The Cirle Surgical Navigation System (Bausch + Lomb) allows for advanced surgical planning and three-dimensional guidance that is shown within the microscope ocular. Active aberrometers, such as Holos (Clarity) and Ora with VerifEye, provide intraoperative refractive measurements that include the effect of posterior corneal curvature on refractive outcomes.
All of these individual technological elements, along with others already available or in development, provide important pieces to an entire system for refining cataract surgery outcomes. What Alcon has accomplished with the latest acquisition of intraoperative aberrometry (Ora with VerifEye) is the summation of a series of integrated, synergistic technologies that allow for optimal surgical outcomes.
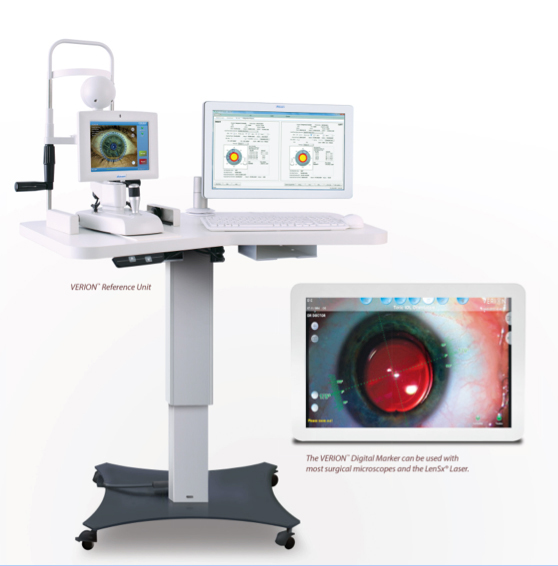
Figure 1
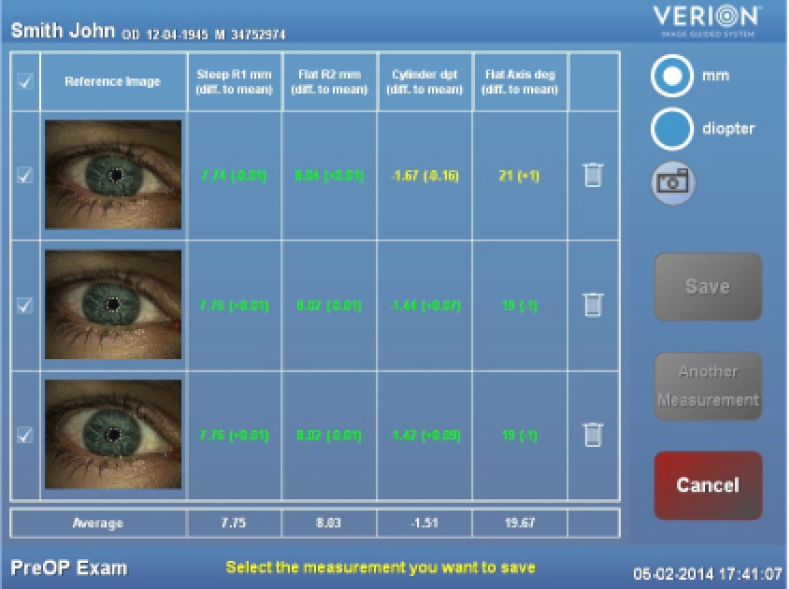
Figure 2
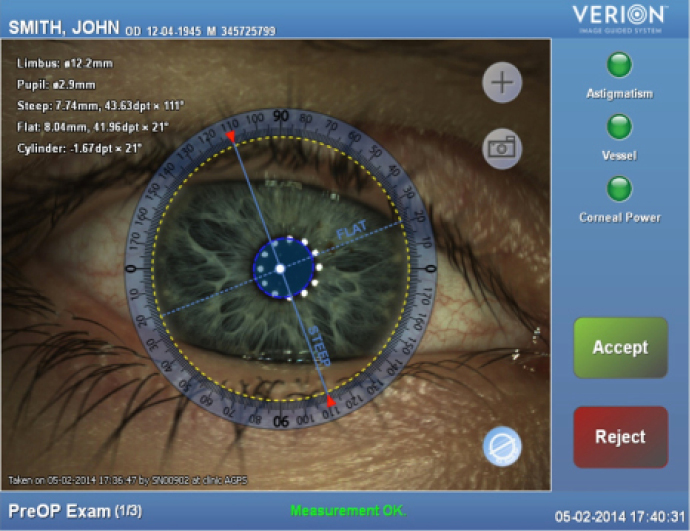
Figure 3
The integrated Alcon Cataract Refractive Suite begins with basic measurement acquisition with the Lenstar. This information can be directly uploaded into the preoperative surgical planner and used to determine the incision placement (including limbal relaxing incisions), lens power, and toric IOL power and orientation (Figures 1 and 2). Preoperative images of the eye are used to register the orientation during subsequent procedures using Verion (Alcon; Figures 3 and 4). This allows for adjustment for cyclorotation both under the LenSx femtosecond laser (Alcon) and in the operating room utilizing the LuxOR LX3 microscope (Alcon; Figure 5). The Centurion phacoemulsification machine (Alcon) integrates with the system as well. The addition of aberrometry will allow for refinement of the plan intraoperatively as well as enable customization of nomograms through individual surgeon data acquisition (Figures 6 and 7).
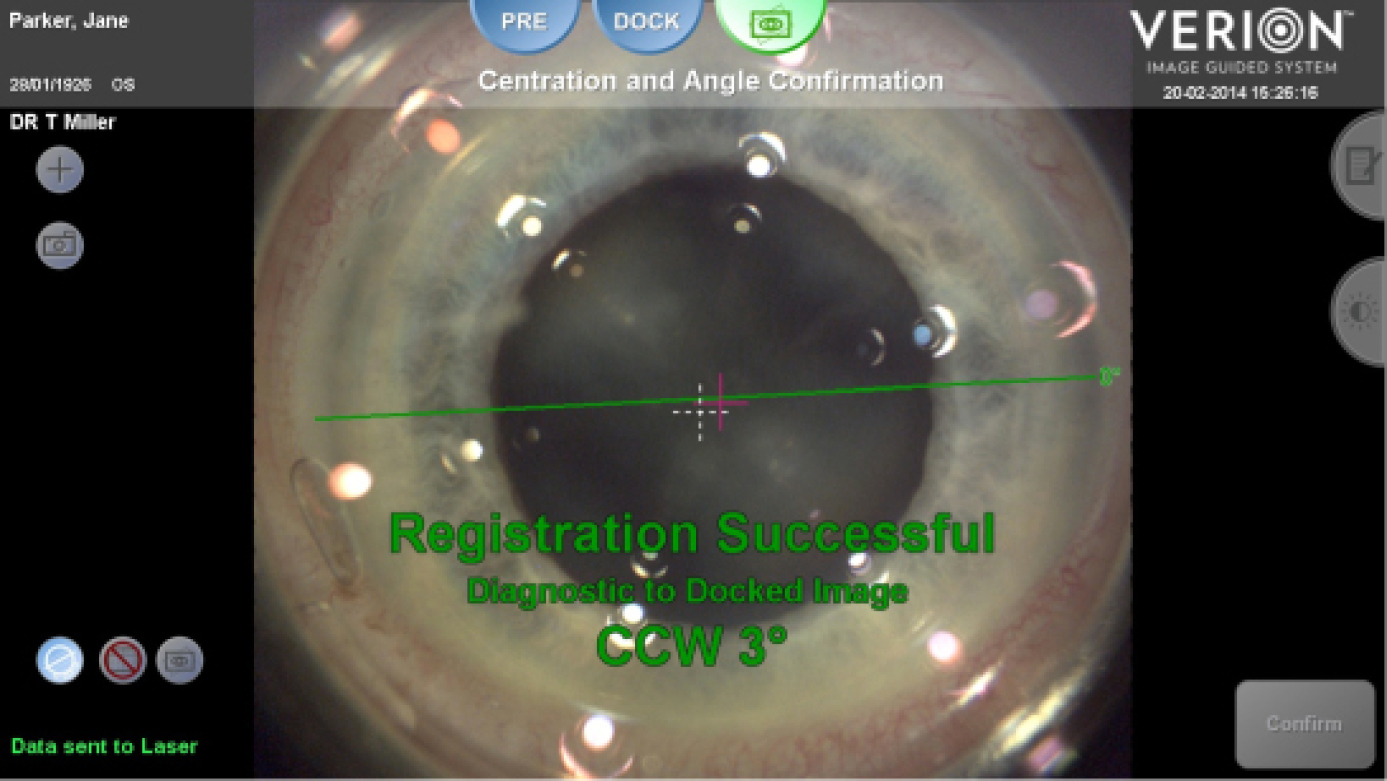
Figure 4
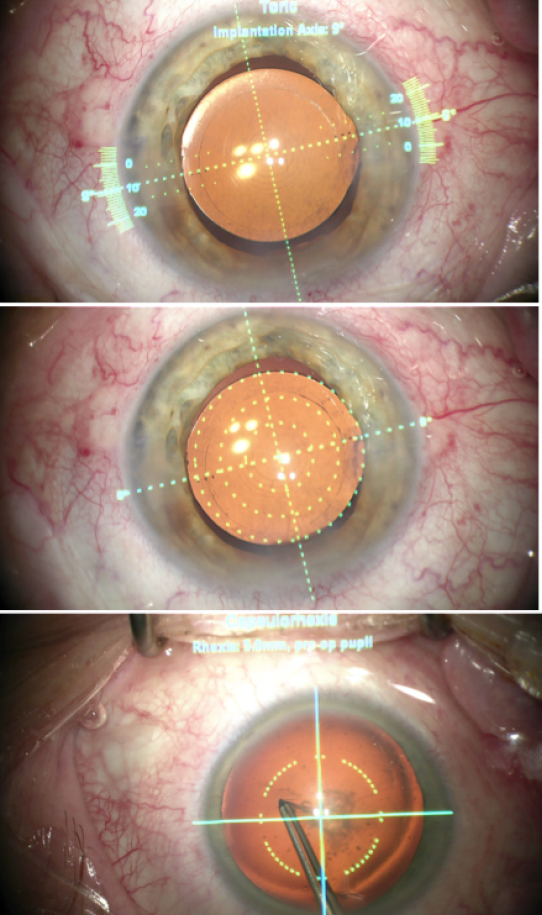
Figure 5

Figure 6
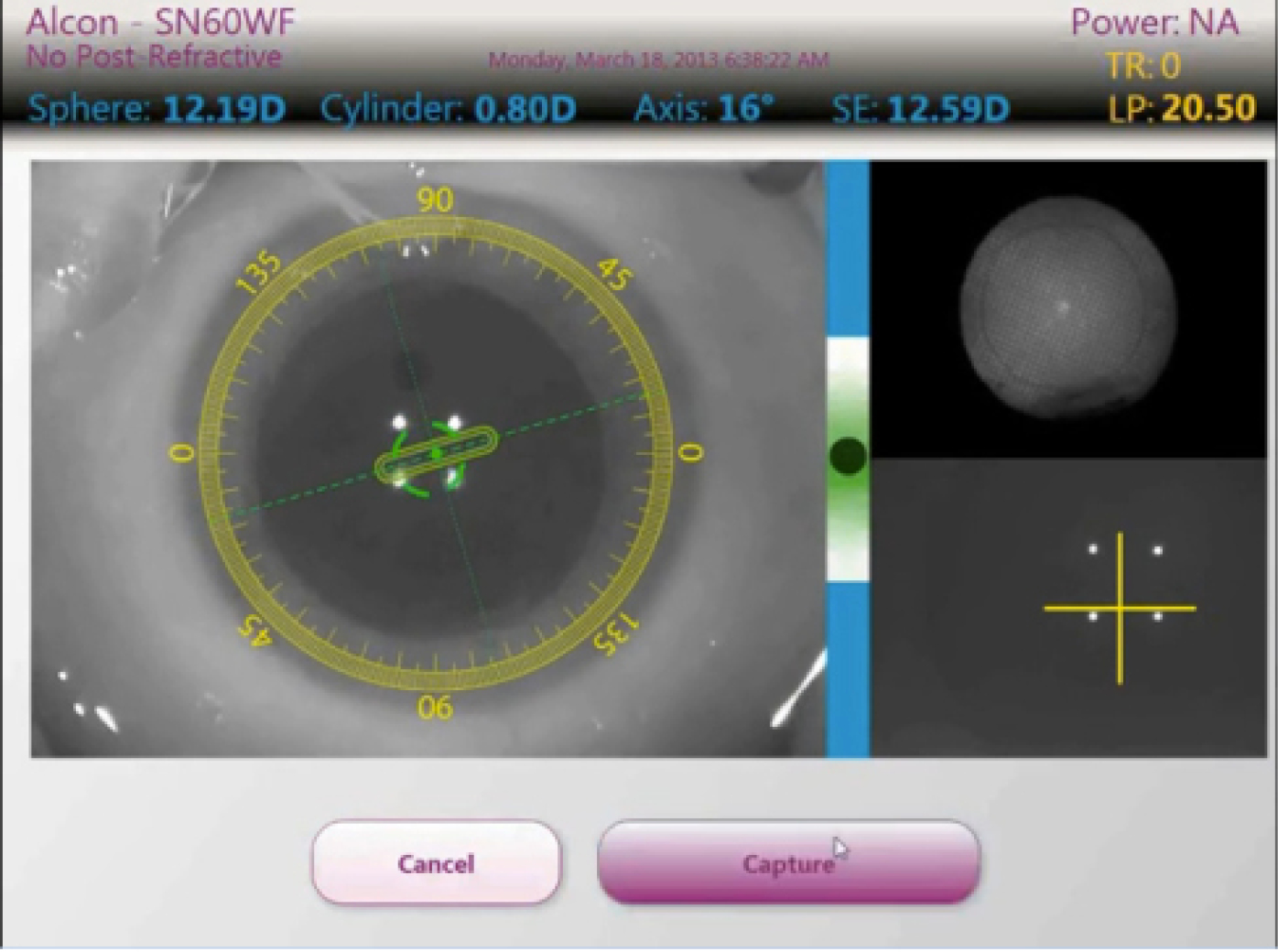
Figure 7
Ultimately, I envision these individual technologies will be combined into a more comprehensive unit that will reside in the operating room and allow for a transition to and from various technologies given the specifics of an individual case, from corneal measurements to surgical planning to intraocular techniques to the femtosecond laser and back again, as needed. All steps will occur with constant real-time feedback regarding the refractive effect of the procedures being performed. Less and less preoperative planning will be needed, as more of the actual measurement and refinement will happen in the operating room. At present, Alcon has the most complete package of technologies to make this integration a reality.
Cathleen M. McCabe, MD, is a cataract and refractive specialist practicing at The Eye Associates in Bradenton and Sarasota, Florida. Dr. McCabe states that she is a speaker for and consultant to Alcon, Bausch + Lomb, and Abbott Medical Optics. She may be reached at (941) 792-2020; cmccabe13@hotmail.com.

Ehsan Sadri, MD, FACS
Cutting-Edge Solutions in Glaucoma and Cataract Surgery
Although there are a variety of game changers in ophthalmology, the two that come to my mind as a glaucoma and cataract surgeon are (1) injectable glaucoma medications and (2) a new class of presbyopia-correcting IOLs. In the glaucoma space, as in other areas of ophthalmology, we face an ongoing issue of patient compliance with their eye drops. The National Eye Institute trials show that patient adherence to medications drops by 50% as an adjunctive medication is added, and thus many patients do not comply with their prescribed drop regimen or do not properly instill their drops, causing them to lose vision. Slow-release injectable glaucoma medications are a potential solution to this issue and could be a real game changer. Allergan is currently conducting a trial exploring the injection of bimatoprost rather than requiring patients to take drops. This could be the next hot thing in ophthalmology, especially if there is a mechanism for surgeons to be able to bill for it. It will allow the practices to absorb the work and create a win-win-win situation for industry, doctors, and patients.

The Symfony has the potential to be a game changer in cataract surgery, as it will mitigate the visual disturbances associated with multifocality.
The other exciting development is in the space of presbyopia-correcting devices—both in the category of IOLs and corneal inlays—such as the new Symfony IOL (Abbott Medical Optics; not available in the United States). The Symfony is an extended range of vision IOL for the treatment of cataract patients with presbyopia that uses a defocus curve and provides a much broader range of vision than a traditional single-focus monovision lens, as demonstrated in early results. The Symfony has the potential to be a game changer in cataract surgery, as it will mitigate the visual disturbances associated with multifocality. Mesopic glare and halos still challenge the multifocal IOLs available for patients, and there is room for major improvement in this sector. Accommodating IOL platforms are good, but patients still seem to require readers for very small print. The cutting-edge design of the Symfony IOL is pretty phenomenal, and the data available thus far look very promising.1-3
Ehsan Sadri, MD, FACS, is a glaucoma and cataract surgeon in practice in Newport Beach, California. Dr. Sadri is a consultant to Abbott Medical Optics and Staar Surgical and a speaker for Allergan. He may be reached at (949) 642-3100; esadrii@gmail.com.
1. New data for Tecnis Symfony presbyopia-correcting extended range of vision IOL presented. EyeWire Today. September 18, 2014. Available at: https://eyewiretoday.com/view.asp?20140918-new_data_for_tecnis_symfony_presbyopia-correcting_extended_range_of_vision_iol_presented. Accessed October 10, 2014.
2. Steinert R, Waltz K, Fajardo-Baide M. Clinical evaluation of a new 1-piece IOL designed to extend range of vision: paired-eye study. Poster presented at: the XXXII Congress of the ESCRS. September 13-17, 2014; London.
3. Hamid A. Early results with the Symfony IOL. Cataract & Refractive Surgery Today Europe. October 2014.

Alice T. Epitropoulos, MD, FACS
Dropless Cataract Surgery
Preventing complications after cataract surgery is always the primary concern for ophthalmic surgeons. For years, cataract surgeons have used drops before and after surgery to prevent inflammation and infection. While these drops are effective, there are disadvantages to using drops perioperatively. A single compounded prophylactic injection is gaining support to avoid having to use drops after cataract surgery.
Patient compliance with drops is a major concern. Many patients forget to use them or administer them as instructed, while others have difficulty instilling drops effectively. In addition, the cost of drops continues to increase due to higher copays and more limited formularies. Some patients therefore choose not to fill their prescriptions altogether, while others, despite physician recommendations, opt for generic drops, which are inferior to branded drops and can carry a higher risk of side effects.
We may now, however, be seeing a game changer enter this space in the form of a single compounded prophylactic injection. Imprimis Pharmaceuticals has developed two proprietary compounded antibiotic and steroid formulations (triamcinolone and moxifloxacin with and without vancomycin) that are administered intrazonularly during cataract surgery, rendering it a “dropless” procedure.
In a retrospective analysis of 738 cataract surgeries in which patients received TriMoxiVanc via a transzonular approach, postoperative drops could be eliminated about 95% of the time.1 A small number of patients in this study had breakthrough inflammation that required initiation of drops. Although surgeons have expressed concern about IOP spikes after these injectable formulations, years of experience with a number of patients have indicated that this is not an issue. In fact, the incidence of IOP elevation seems to be more prevalent with postoperative topical steroids than with intravitreal TriMoxi and TriMoxiVanc injections. There were no cases of pressure spikes or endophthalmitis observed in the retrospective analysis.
Intraoperative administration of the Imprimis formulations eliminates both patient error and noncompliance associated with drops. The injection also reduces the chance of causing corneal toxicity or exacerbating ocular surface disease from preservatives in topical drops. Further, as the cost of eye drops has become more of a concern for many patients, the Imprimis injection is extremely cost effective, ranging from $20 to $25. Surgeons using the dropless formulations will quickly find that their staff members appreciate not getting pharmacy phone calls, insurance authorization forms, and patient complaints about price versus copays.
In addition, these formulations can be used in patients with diabetic retinopathy, known preoperative diabetic macular edema, epiretinal membranes, vitreomacular adhesion and vitreomacular traction, age-related macular degeneration, or any other maculopathy. Also, because IOP has not been an issue, glaucoma patients, ocular hypertensives, and glaucoma suspects can also receive the injectable medication.
As with any medication, TriMoxi can have side effects, including peripheral floaters and blurred vision in the immediate postoperative period; however, the floaters appear to be transient, clearing within a few days after surgery. Although some surgeons may be concerned about the use of compounded medications, Imprimis has addressed this with strict adherence to quality control and sterility protocols. Immediately after the company’s first acquisition of a prescription fulfillment pharmacy, it began implementing internal quality assurance standards and best practice policies that exceed those required under the US Pharmacopeia and state pharmacy laws.
Compounded injectable medications are revolutionizing cataract surgery. They simplify the process and create a better atmosphere for the patient and staff both pre- and postoperatively. Patient noncompliance and potential for error with drops can be eliminated while inflammation and infection is prevented.
Alice T. Epitropoulos, MD, FACS, is a cataract and refractive surgeon at The Eye Center of Columbus and a Clinical Assistant Professor at The Ohio State University Wexner Medical Center. Dr. Epitropoulos states that she has no financial interest in the products or companies mentioned. She may be reached at epitrop@columbus.rr.com.
1. Liegner JT. Intravitreal antibiotics and steroid for dropless cataract surgery. Presented at: the American Society of Cataract and Refractive Surgery Annual Meeting; April 27, 2014; Boston, MA.
