Endothelial cell density is a major consideration in the preoperative workup and postoperative care of patients undergoing corneal and cataract surgery. For example, this information is key to assessing the risk of corneal decompensation after intraocular surgery. Cell counts allow surgeons to plan protective intraoperative measures for patients undergoing a cataract procedure or IOL exchange, and they aid candidate selection for a phakic IOL. Cell counts also allow surgeons to monitor the health of corneal grafts, and they contribute to the detection of problems following the placement of a phakic IOL.
At a Glance
- TheEndothelial cell density is a major consideration in the preoperative workup and postoperative care of patients undergoing corneal and cataract surgery.
- TheThe field of view with current high-resolution clinical modalities is limited. Although some research devices offer wider fields of view, they are subject to motion artifacts or capture flat optical sections of the sample, which limits the view of the naturally curved cornea.
- TheCurved-field OCT may surmount these limitations.
The field of view with current high-resolution clinical modalities such as in vivo confocal microscopy and specular microscopy is limited to approximately 0.5 mm. Several in vivo research devices based on Fourier-domain OCT that are in development offer larger fields of view, but motion artifacts are common unless the subject is anesthetized (ie, immobilized). A recently reported method of in vivo time-domain full-field OCT can obtain cell-detail corneal images with a viewer area that is 10 times larger than the clinical state of the art, but, like confocal microscopy, it captures flat optical sections of the sample.1,2 This limits the view of the naturally curved cornea because the flat field optically sections through several corneal layers at once.
Researchers may now have developed an approach that surmounts these limitations: curved-field OCT (CF-OCT).3
METHOD
According to Mazlin and colleagues,1-3 CF-OCT can capture optical sections of arbitrarily selected curvature, which allows optical flattening of curved samples in vivo (Figure 1). Applied to the cornea, CF-OCT can provide a 1.13 x 1.13-mm view of the curved subbasal nerve plexus and layers of the corneal endothelium (Figure 2). The method’s high en face imaging speed (0.6 billion pixels/s) reportedly eliminates artifacts from movements of the eye or head. CF-OCT is a noncontact method that takes 3.5 milliseconds to execute.

Figure 1 | To capture images from a curved sample such as a cornea, Mazlin and colleagues replaced the flat mirror (A) of a traditional full-field OCT setup with a curved optical lens (B). The curved lens optically flattens the acquired images.
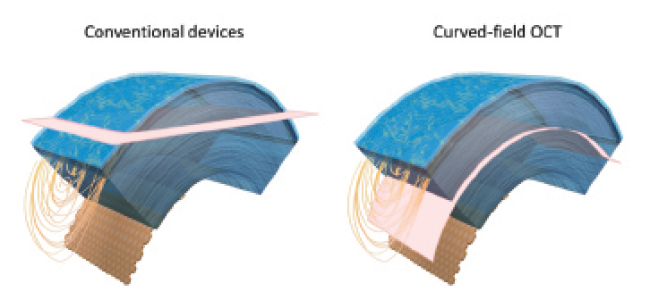
Figure 2 | Traditional imaging approaches acquire a flat slice (pink) that crosses through several corneal layers at once, limiting the field of view (A). CF-OCT matches the curvature of the cornea to provide a larger imaging area (B).
“If you’re looking ... at a given cell layer like the endothelial layer of the cornea or if you want to look at the corneal nerve structure en face, this technology allows you to do that with a very, very fast capture of the image,” Joel S. Schuman, MD, remarked to BMC Vision. Dr. Schuman, Professor and Vice Chairman for Ophthalmology Research at NYU Langone Health in New York, is one of the inventors of OCT.
In a quantitative comparison, Mazlin and colleagues3 counted 1,743 cells over a 0.85-mm–diameter circular area of best focus within the CF-OCT field; this was equivalent to a cell density of 3,072 cells/mm2—a normal count for a human cornea. With specular microscopy, they counted 387 cells and measured a cell density of 3,096 cells/mm2.
ADVANTAGES
CF-OCT is very high resolution, both axially and transversely. That’s a big advantage over other modalities, according to Dr. Schuman—especially for evaluating the endothelium, anterior corneal layers, and corneal nerves. A scanning laser ophthalmoscope, for example, achieves very high transverse resolution but lower axial resolution, he noted. Moreover, the view encompasses multiple layers of tissue, whereas CF-OCT can image a single layer.
Although the field of view with CF-OCT is larger than with clinically available modalities, it is still relatively limited. In other words, “it’s not like being able to look at the entire cornea,” Dr. Schuman said. “On the other hand, what you’re able to see is quite impressive.”
FUTURE DIRECTIONS
The current study by Mazlin and colleagues focuses on the use of CF-OCT for anterior segment applications, but the investigators state that this method may have utility for monitoring the progression of diabetes.3 Additional applications may be developed over time.
One issue with the current device that Mazlin and colleagues identified is that changing the curvature of optical sectioning requires changing a lens and performing a realignment. The researchers note that it may be possible to increase the flexibility of future iterations of the device through the use of static and tunable lenses. The incorporation of automatic cell-counting techniques could also be beneficial.
1. Mazlin V, Xiao P, Scholler J, et al. Real-time non-contact cellular imaging and angiography of human cornea and limbus with common-path full-field/SD OCT. Nat Commun. 2020;11(1):1868.
2. Mazlin V, Xiao P, Dalimier E, et al. In vivo high resolution human corneal imaging using full-field optical coherence tomography. Biomed Opt Express. 2018;9(2):557-568.
3. Mazlin V, Irsch K, Paques M, Sahel JA, Fink M, Boccara CA. Curved-field optical coherence tomography: large-field imaging of human corneal cells and nerves. Optica. 2020;7(6):872-880.


