By 2020, there will be 2.1 billion presbyopes worldwide, many of whom desire spectacle independence. The two main surgical approaches to treat presbyopia in patients aged 40 to 50 years are corneal refractive surgery and refractive lens exchange.1 Laser vision correction to create a monovision target, or a multifocal ablation can be performed; however, side effects include reduced distance visual acuity, stereopsis, contrast sensitivity, and quality of vision. Lenticular intraocular surgeries with multifocal and accommodating lenses or a monovision target are invasive and have associated risks, such as intraocular complications, biometric errors, reduced contrast sensitivity, and loss of BCVA.2 More recently, there has been a significant interest in corneal inlays because they are an additive technology and do not require any tissue removal.1,2
The Flexivue Microlens, Raindrop Near Vision Inlay, and Kamra Inlay.
Currently, three corneal inlays commercially available or in development for the treatment of presbyopia include: (1) the Flexivue Microlens (Presbia), which changes the refractive index by means of a bifocal optic; (2) the Raindrop Near Vision Inlay (ReVision Optics), which reshapes the anterior corneal curvature; and (3) the Kamra inlay (AcuFocus), which utilizes pinhole optics to increase the eye’s depth of focus. The Kamra recently received FDA approval in April of this year.
FLEXIVUE MICROLENS
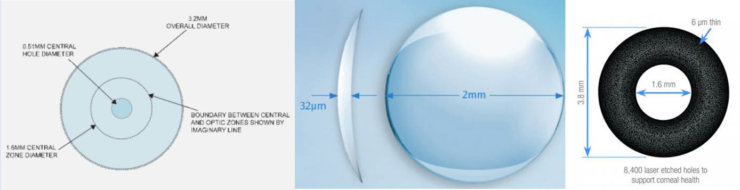
The Flexivue Microlens is a clear hydrogel implant that is 3 mm in diameter and has a central plano zone for distance vision, surrounded by rings of varying refractive power (+1.25 to +3.00 in 0.25-D increments) for intermediate and near vision. The implant is 15 μm thick and has a 0.15-mm central opening to facilitate nutrient and oxygen transport. It is inserted in a corneal pocket at a depth of 280 to 300 μm.
Limnopoulou et al3 reported on the refractive outcomes of 47 patients implanted with the Flexivue Microlens in the nondominant eye with 1-year follow-up. Mean near UCVA was 20/32 or better in 75% of patients (mean 20/25). Monocular mean distance UCVA decreased from 20/20 to 20/50 but was not affected binocularly, and 37% of patients lost one line of BCVA. There was a decrease in contrast sensitivity and an increase in higher-order aberrations, but overall, patient satisfaction was high.3 The Flexivue Microlens is currently being tested in the United States.
RAINDROP
The Raindrop Near Vision Inlay is a clear hydrogel, highly permeable implant that is 2 mm in diameter and has a thickness of 32 μm centrally and 10 μm peripherally. The Raindrop does not have intrinsic refractive power, as it has a similar refractive index as the cornea; it enhances intermediate and near vision by increasing the anterior curvature of the cornea and creating a hyperprolate shape, resulting in a multifocal cornea. It is intended for implantation under a 130-μm to 150-μm LASIK flap or in a corneal pocket in the nondominant eye.
Garza et al4 implanted the Raindrop in 19 presbyopic eyes and followed the visual outcomes for 1 year. One hundred percent of patients achieved a near UCVA of 20/25 or better, and mean binocular distance UCVA was 20/20 or better. Mean contrast sensitivity was only slightly decreased. Ninety-five percent of patients reported that they were either satisfied or very satisfied with their near and intermediate vision, and all were very satisfied with distance vision. Eighty-four percent of patients reported that they rarely used glasses. One patient, however, required removal of the device due to dissatisfaction, and another required recentration.4
Chayet et al5 published their results of 16 hyperopic presbyopic patients who had LASIK concurrently with Raindrop insertion in the nondominant eye. After 1 year, mean near UCVA was 20/21, mean intermediate UCVA was 20/26, and distance UCVA was 20/31. Binocularly, near UCVA, intermediate UCVA, and distance UCVA were 20/21, 20/26, and 20/19, respectively. On the patient questionnaire, 14 out of 16 patients reported being satisfied or very satisfied with their vision, with minimal reports of glare and halos. One device was explanted secondary to haze formation.5 The Raindrop device is currently in clinical trials in the United States.
KAMRA
The Kamra inlay is a perforated opaque inlay made of polyvinylidene fluoride, with incorporated nanoparticles of carbon. The device is 5 μm thick and 3.8 mm in diameter, with a central aperture of 1.6 mm. It has 8,400 perforations to facilitate oxygen and nutrient flow within the cornea. The Kamra relies on pinhole optics to block unfocused light (6.7% transmission of light through the opaque part of the inlay), which results in an increase in depth of focus and improvement in near and intermediate vision. It is implanted in a corneal pocket created by a femtosecond laser at a depth of 180 μm or greater in the nondominant eye. Tabernero and Artal6 have suggested that the best depth of focus can be obtained by leaving patients with a residual myopia of -0.50 to -0.75 D in the Kamra-implanted eye.
The Kamra has been the most studied inlay in the literature, with more than 20,000 inlays implanted worldwide and approval in 51 countries, including the recent FDA approval. As a result, it has been extensively studied in virgin and post-LASIK emmetropes, in conjunction with LASIK correction as a simultaneous or two-step procedure and in pseudophakes after implantation of a monofocal IOL.1 Long-term studies show the device to be safe, with no inflammatory reactions, stromal fibrosis, or significant change in endothelial cell count. Central and peripheral iron deposits occurred in more than 56% of eyes implanted with the first generation of this inlay, but a much lower incidence was seen with the newer model, which is thinner, implanted more deeply in the cornea, and has less light transmission (6.7% vs 7.5%).
Tomita et al7 published the largest report on the Kamra, in which 223 patients with previous LASIK had the current inlay implanted. Mean UDVA decreased from 20/16 to 20/20 at 6 months postoperatively. Mean UNVA was J2, and there was a significant increase in the spectacle independence for reading and great patient satisfaction.7 Dexl et al8 published the study with the longest follow-up—5 years—in 32 emmetropic presbyopic patients. Despite the fact that the study was done using the older Kamra model, the study showed the device to be effective and safe. At 5 years, 74.2% of patients had a near UCVA of J3 or better, 87.1% had an intermediate UCVA of 20/32 or better, and 93.5% had a distance UCVA of 20/20 or better. Four complications occurred: removal of one implant secondary to patient dissatisfaction, two recentrations, and one episode of epithelial ingrowth.8
No visual field scotomas were noted after implantation of the Kamra inlay.1 In addition, presbyopes improved on all measures of reading performance.9 Patient satisfaction with UCVA has been high, especially in emmetropes and hyperopes.1,10 In a recent study, Vilupuru et al11 showed that patients implanted with the Kamra device had better contrast sensitivity compared with patients implanted with the Tecnis Multifocal (Abbott Medical Optics) or the Restor IOL (Alcon). In addition, both the Crystalens (Bausch + Lomb) and the Kamra had better intermediate UCVA than the multifocal lenses.11 No significant modifications were required to safely perform cataract surgery 4 years after Kamra inlay implantation in two patients. The biometry readings were satisfactory, UCVA was good, and near UCVA was 20/20 following cataract surgery.12
CONCLUSION
No study published any serious sight-threating complications, although patients have had minimal complaints of glare and halos, dry eyes, and night vision problems. Management of dry eye is paramount in achieving good results. Centration on the visual axis is also very important to achieve the best refractive outcomes, as decentration of as little as 0.5 mm can significantly reduce image quality.10
Although there are some rare complications, corneal inlays are generally well tolerated and associated with high patient satisfaction. It is exciting to provide a technology for treating presbyopia without greatly compromising distance vision. In addition, these inlays can be removed if necessary, with no permanent effect on corneal topography and aberrometry and no difference in distance BCVA pre- and postoperatively.13 With the recent FDA approval of the Kamra inlay, I am eager to start implanting these devices. In the future, there will most likely be studies to directly compare the devices and to improve patient selection.
1. Lindstrom RL, Macrae SM, Pepose JS, Hoopes PC Sr. Corneal inlays for presbyopia correction. Curr Opin Ophthalmol. 2013;24(4):281-287.
2. Arlt E, Krall E, Moussa S, Grabner G, Dexl A. Implantable inlay devices for presbyopia: the evidence to date. Clin Ophthalmol. 2015;9:129-137.
3. Limnopoulou AN, Bouzoukis DI, Kymionis GD, et al. Visual outcomes and safety of a refractive corneal inlay for presbyopia using femtosecond laser. J Refract Surg. 2013;29(1):12-18.
4. Garza EB, Gomez S, Chayet A, Dishler J. One-year safety and efficacy results of a hydrogel inlay to improve near vision in patients with emmetropic presbyopia. J Refract Surg. 2013;29(3):166-172.
5. Chayet A, Barragan Garza E. Combined hydrogel inlay and laser in situ keratomileusis to compensate for presbyopia in hyperopic patients: one-year safety and efficacy. J Cataract Refract Surg. 2013;39(11):1713-1721.
6. Tabernero J, Artal P. Optical modeling of a corneal inlay in real eyes to increase depth of focus: optimum centration and residual defocus. J Cataract Refract Surg. 2012;38(2):270-277.
7. Tomita M, Kanamori T, Waring GO, Nakamura T, Yukawa S. Small-aperture corneal inlay implantation to treat presbyopia after laser in situ keratomileusis. J Cataract Refract Surg. 2013;39(6):898-905.
8. Dexl AK, Jell G, Strohmaier C, et al. Long-term outcomes after monocular corneal inlay implantation for the surgical compensation of presbyopia. J Cataract Refract Surg. 2015;41(3):566-575.
9. Dexl AK, Seyeddain O, Riha W, et al. Reading performance and patient satisfaction after corneal inlay implantation for presbyopia correction: two year follow-up. J Cataract Refract Surg. 2012;38:1808-1816.
10. Tomita M, Kanamori T, Waring GO 4th, et al. Simultaneous corneal inlay implantation and laser in situ keratomileusis for presbyopia in patients with hyperopia, myopia, or emmetropia: six-month results. J Cataract Refract Surg. 2012; 38:495-506.
11. Vilupuru S, Lin L, Pepose JS. Comparison of contrast sensitivity and through focus in small-aperture inlay, accommodating intraocular lens, or multifocal intraocular lens subjects. Am J Ophthalmol. 2015;160(1):150-162.
12. Tan TE, Mehta JS. Cataract surgery following KAMRA presbyopic implant. Clin Ophthalmol. 2013;7:1899-1903.
13. Alió JL, Abbouda A, Huseynli S, Knorz MC, Homs ME, Durrie DS. Removability of a small aperture intracorneal inlay for presbyopia correction. J Refract Surg. 2013;29(8):550-556.





