According to the World Health Organization’s latest assessment (2010), cataracts cause 51% of blindness worldwide. At that time, therefore, approximately 20 million people were affected by cataract.1 Today, that number is undoubtedly larger. It is therefore not surprising that cataract surgery is one of the most commonly performed procedures in the world, with Market Scope predicting a steady growth in volume over the next 5 years.2
Thanks to technical and technological advances, the procedure has become increasingly successful, yet a source of frustration for surgeons and their affected patients persists: dysphotopsias. Hu and colleagues recently reviewed the literature on this subject to summarize current understanding of these phenomena, their prevention, and their management.3 ME invited several experts in this area to share their thoughts on the review, what is known about dysphotopsias today, and what answers are still being sought.
—Gillian McDermott, MA, Editor-in-Chief, Clinical Content, Anterior Segment, BMC Vision
JACK T. HOLLADAY, MD, MSEE, FACS
The review by Hu and colleagues3 ignores entoptic phenomena, but the distinction between those and dysphotopsias is one worth making. Together, the two categories account for the breadth of unwanted visual symptoms patients may experience after cataract surgery (Figure 1).
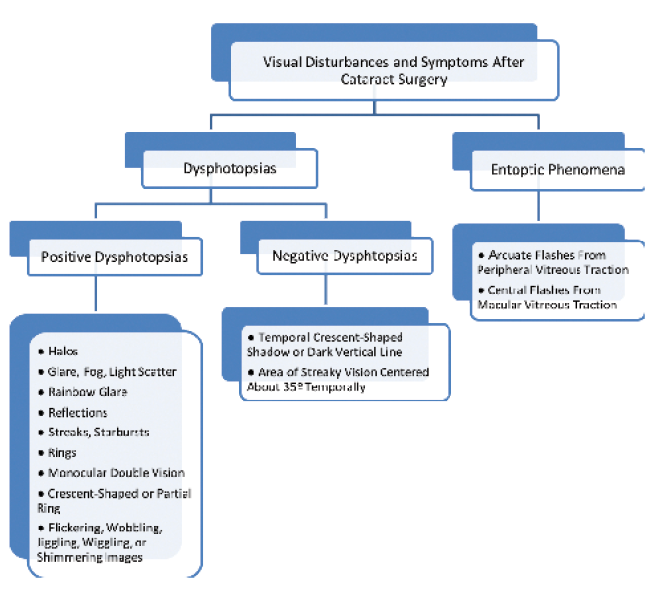
Figure 1 | Unwanted visual symptoms that patients may experience after cataract surgery.
(Courtesy of Jack T. Holladay, MD, MSEE, FACS)
Dysphotopsias require some sort of stimulus from outside the eye, and they may be categorized as positive (PD) and negative (ND) dysphotopsias, which have been described in detail optically.4,5 In contrast, the source of entoptic phenomena is within the eye itself. Usually, entoptic phenomena result from partial peripheral or central vitreous collapse, which puts traction on the retina and results in the perception of light without any light stimulus. The flashes occur with the eye closed in complete darkness as the head moves quickly from side to side or up and down. Entoptic phenomena have nothing to do with the IOL itself, but they frequently occur immediately after cataract surgery because the IOL occupies much less volume in the eye than did the crystalline lens, leaving more of the posterior compartment for the existing vitreous.
A knowledge of the categories of unwanted visual phenomena helps surgeons properly diagnose and manage the problem postoperatively.
JAMES A. DAVISON, MD
I commend Hu and colleagues3 on their thorough review of pseudophakic dysphotopsia. There were two reasons for categorizing dysphotopsias into PD and ND. First, it seemed that there must be different etiologies because the symptoms of PD and ND are completely different, as are the clinical courses and responses to treatment. Second, both problems seemed to require an anomalous vulnerability of the vision system for symptoms to become significantly manifest.
It seems that PD is associated with IOLs made of high refractive index acrylic material that have a relatively flat anterior power curve and tall, square-edged optics. Symptoms usually do not improve over time, and exchanging the IOL for one with opposite design features or with significantly different characteristics seems to resolve the issue. To prevent the problem, I recommend choosing IOLs without all or at least without some of the aforementioned PD-associated characteristics, especially the flatter anterior surface and high refractive index plastic, for patients who have modest nuclear cataracts, substantial complaints of glare before surgery, and a lifelong history of difficulty with driving at night. These individuals account for about 3% of patients in my practice. I do not recommend multifocal IOLs for these patients, but I will use toric IOLs after some extra admonitions during informed consent.
Because of its almost always immediate resolution with secondary reverse optic capture, ND seems to be most associated with nasal anterior capsular overlap, but the phenomenon is also more frequently seen in patients who received an IOL with a square-edged optic that is made of high refractive index plastic. ND can be observed in patients who have no nasal anterior capsular overlap and also has been resolved by amputation of the far nasal optic. Small degrees of ND are commonly experienced right after surgery. These observations are usually declared almost parenthetically and are so common that they might be considered a normal side effect because they almost always resolve spontaneously. I would liken it to the feeling of stiffness in a new joint after total knee replacement surgery. Mild symptoms almost always resolve within a month or 2. The more severe the symptoms are, the more likely they are to persist.
Clinical perspective is important. In an unpublished study, I prospectively catalogued instances of significant and persistent dysphotopsia from 2010 to 2015 and logged only 21 (12 PD and nine ND) occurrences of symptoms that might merit reoperation out of 14,726 surgeries (0.16%). About half in each category required surgery, which cured most of the patients completely or almost completely.
After 69 years of evolution, the appearance of modern prosthetic lenses hardly imitates that of the natural lens. It is a wonder that IOLs work as well as they do. That said, thanks to Sir Harold Ridley and countless other surgeons and engineers, cataract surgery with IOL implantation continues to safely restore vision to millions of patients around the world every year. All things considered, it is one of the best success stories in medicine today, and I look forward to the availability of new IOL designs, particularly the one being developed by Samuel Masket, MD (see next section), which should reduce patients’ risk of both PD and ND.
SAMUEL MASKET, MD; NICOLE R. FRAM, MD; AND ZSÓFIA RUPNIK, MD
Hu and colleagues3 provided an excellent summary of the dysphotopsia literature through September 2017. In their comprehensive, albeit somewhat short, article they included PD, ND, multifocal dysphotopsia, and ring dysphotopsia. Of these, ND remains the least well-understood condition. Unfortunately, our report on the largest series of eyes to date requiring surgery for chronic ND and the rationale behind the surgical strategies was not included because it was published in January 2018.6 In that research, we believe that we uncovered several myths about ND, and we attempted to dispel them in the report.
One such myth is that ND is induced primarily by square-edged hydrophobic acrylic IOLs that have a high index of refraction. Of 40 eyes requiring surgery for ND in our study, 77% had acrylic IOLs and 23% had silicone IOLs. More to the point, 13% of the lenses had round edges, and all cases involved IOLs placed in the bag with an overlying anterior capsulotomy. That last finding is true across all literature regarding ND. An earlier study by Burke and Benjamin amplified the concept; removing various IOLs from the capsular bag and replacing them with high-index acrylic IOLs in the sulcus cured all five cases of ND.7 Similarly, Vámosi et al demonstrated a cure of ND when IOLs were shifted from the bag to the sulcus, irrespective of IOL material.8 We also believe that increased posterior chamber depth is not a primary causative factor, given that Nd:YAG laser removal of the nasal capsule and removal of the nasal aspect of an IOL in the bag have been reported to relieve ND without altering the IOL position.9-11 It is clear to us that ND is enigmatic in many aspects and that the etiology is likely multifactorial, but evidence firmly indicates that ND is associated with any in-the-bag IOL with an overlying continuous circular anterior capsulotomy.
Consistent with our original concept, we found reverse (anterior) optic capture to be a highly successful remedial and prophylactic strategy.6,12 One of us (SM) has designed an antidysphotopic IOL (Masket ND IOL Type 90S, Morcher) that received the CE Mark and is in clinical trials in Europe; the device appears to be highly successful in preventing ND.2
ND remains difficult to investigate because there are no truly objective means by which to assess patient observations. Researchers have used ray tracing to help understand the etiology of ND and the contribution of different IOL characteristics, but the results of those investigations have not been fully correlated or consistent with the clinical manifestations of ND.
PÉTER VÁMOSI, MD, PhD, Med Habil
I agree with Hu and colleagues3 that PD is better understood than ND, but in the past few years more information has become available about the latter. I am particularly interested in ND research. One of my patients was an architect, and he illustrated the shadow he observed with his right eye in different positions of gaze. In nasal gaze, the shadow associated with ND became wider, whereas in temporal gaze the shadow almost completely disappeared (Figure 2).

Figure 2 | Depiction of ND in different positions of gaze by one of Dr. Vámosi’s patients.
(Courtesy of Péter Vámosi, MD, PhD, Med Habil)
In my everyday practice, I try to calm affected patients and advise them to do nothing in the first 6 months because there is a real chance of spontaneous resolution. After that, I suggest step-by-step therapy. It starts with nasal reverse optic capture or with the placement of a piggyback IOL, followed by an IOL exchange in which I place a three-piece posterior chamber IOL with a round anterior edge in the ciliary sulcus.
Primary reverse optic capture is one method by which to prevent ND in a patient’s second operated eye. Recently, I participated in a study of the Masket ND IOL Type 90S, which was developed for the prevention of ND. The IOL has two conventional haptics for fixation in the bag and a circumferential groove on the optic edge for capsulorhexis fixation. The goal of the groove is to hinder ND by controlling the interaction between the anterior capsule and the optic edge. Of the 61 patients in whom I implanted this IOL, none experienced ND, although three had previously experienced ND after earlier surgery in their fellow eye with implantation of another type of IOL.
1. World Health Organization. Blindness and vision impairment prevention. https://bit.ly/1rTbp2S. Accessed July 25, 2018.
2. Steady growth in cataract surgical procedures is expected over the next 5 years. Eyewire News. July 27, 2017. https://bit.ly/2NKR3Yy. Accessed July 25, 2018.
3. Hu J, Sella R, Afshari NA. Dysphotopsia: a multifaceted optic phenomenon. Curr Opin Ophthalmol. 2018;29:61-68.
4. Holladay JT, Lang A, Portney V. Analysis of edge glare phenomena in intraocular lens edge designs. J Cataract Refract Surg. 1999;25(6):748-752.
5. Holladay JT, Simpson MJ. Negative dysphotopsia: causes and rationale for prevention and treatment. J Cataract Refract Surg. 2017;43(2):263-275.
6. Masket S, Fram NR, Cho A, et al. Surgical management of negative dysphotopsia. J Cataract Refract Surg. 2018;44(1):6-16.
7. Burke TR, Benjamin L. Sulcus-fixated intraocular lens implantation for the management of negative dysphotopsia. J Cataract Refract Surg. 2014;40(9):1469-1472.
8. Vámosi P, Csákány B, Németh J. Intraocular lens exchange in patients with negative dysphotopsia symptoms. J Cataract Refract Surg. 2010;36(3):418-424.
9. Folden DV. Neodymium:YAG laser anterior capsulectomy: surgical option in the management of negative dysphotopsia. J Cataract Refract Surg. 2013;39(7):1110-1115.
10. Cooke DL, Kasko S, Platt LO. Resolution of negative dysphotopsia after laser anterior capsulotomy. J Cataract Refract Surg. 2013;39(7):1107-1109.
11. Alapati NM, Harocopos GJ, Sheybani A. In-the-bag nasal intraocular lens optic truncation for treatment of negative dysphotopsia. J Cataract Refract Surg. 2016;42(12):1702-1706.
12. Masket S, Fram N. Pseudophakic negative dysphotopsia: surgical management and new theory of etiology. J Cataract Refract Surg. 2011;37(7):1199-1207.








